Details of Drug-Drug Interaction
| Drug General Information (ID: DDITDQAGB4) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Zoledronic acid | Drug Info | Thalidomide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Bone Density Conservation Agents | Immunosuppressive Agents | |||||||
| Structure | |||||||||
| Mechanism of Zoledronic acid-Thalidomide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
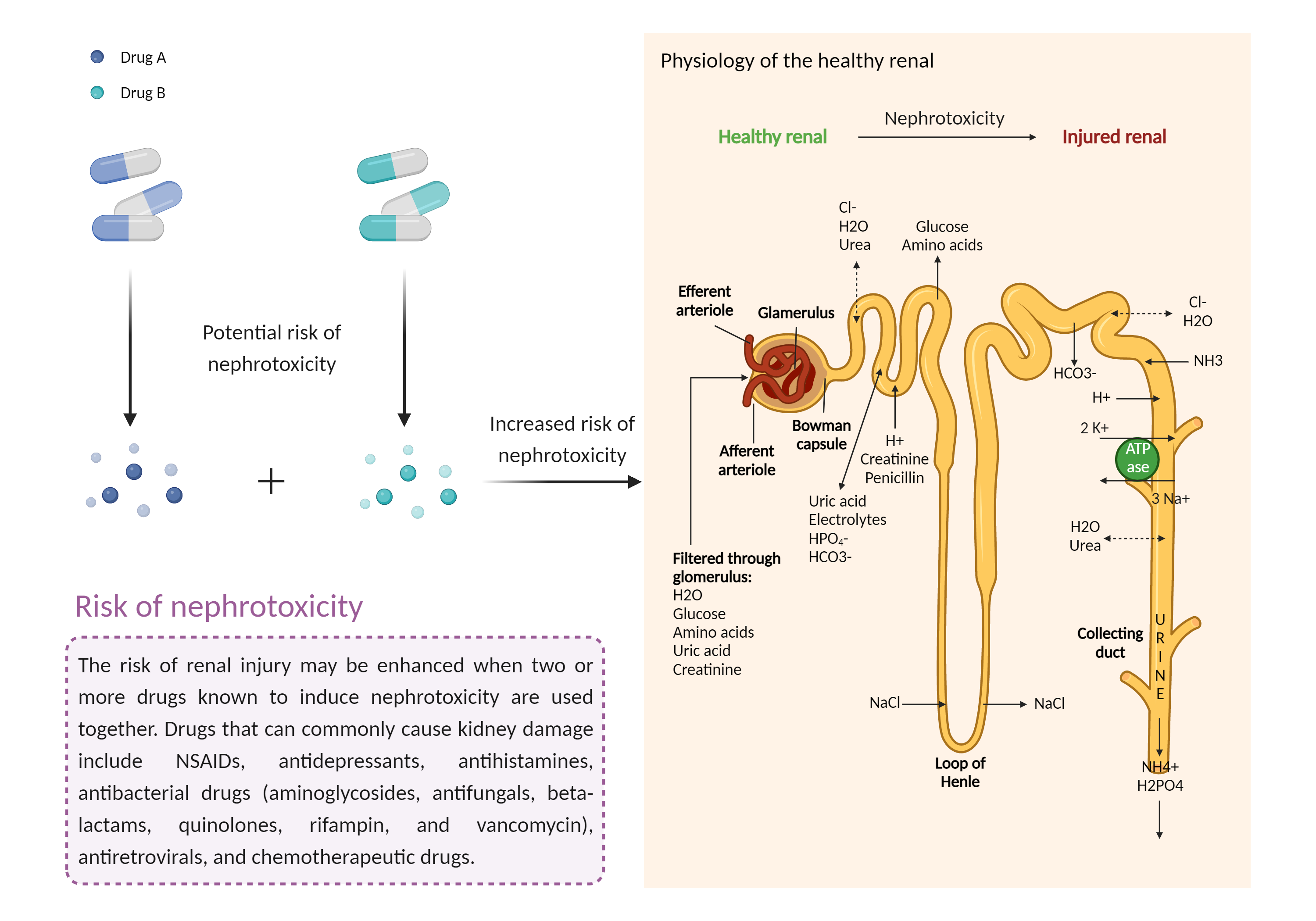
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Zoledronic acid | Thalidomide | |||||||
| Mechanism | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if pamidronate, zoledronic acid, or other intravenous formulations of bisphosphonates must be used in multiple myeloma patients who are receiving treatment with thalidomide. Renal function and serum electrolytes should be monitored. Product labelings for pamidronate and zoledronic acid recommend that single doses not exceed 90 mg and 4 mg, respectively, and the duration of infusion not less than 2 hours and 15 minutes, respectively. Patients should have serum creatinine assessed prior to each treatment, and treatment should be withheld for renal deterioration. In those treated for bone metastases, treatment should not be resumed until renal function returns to baseline. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Zometa (zoledronic acid). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
