| Management |
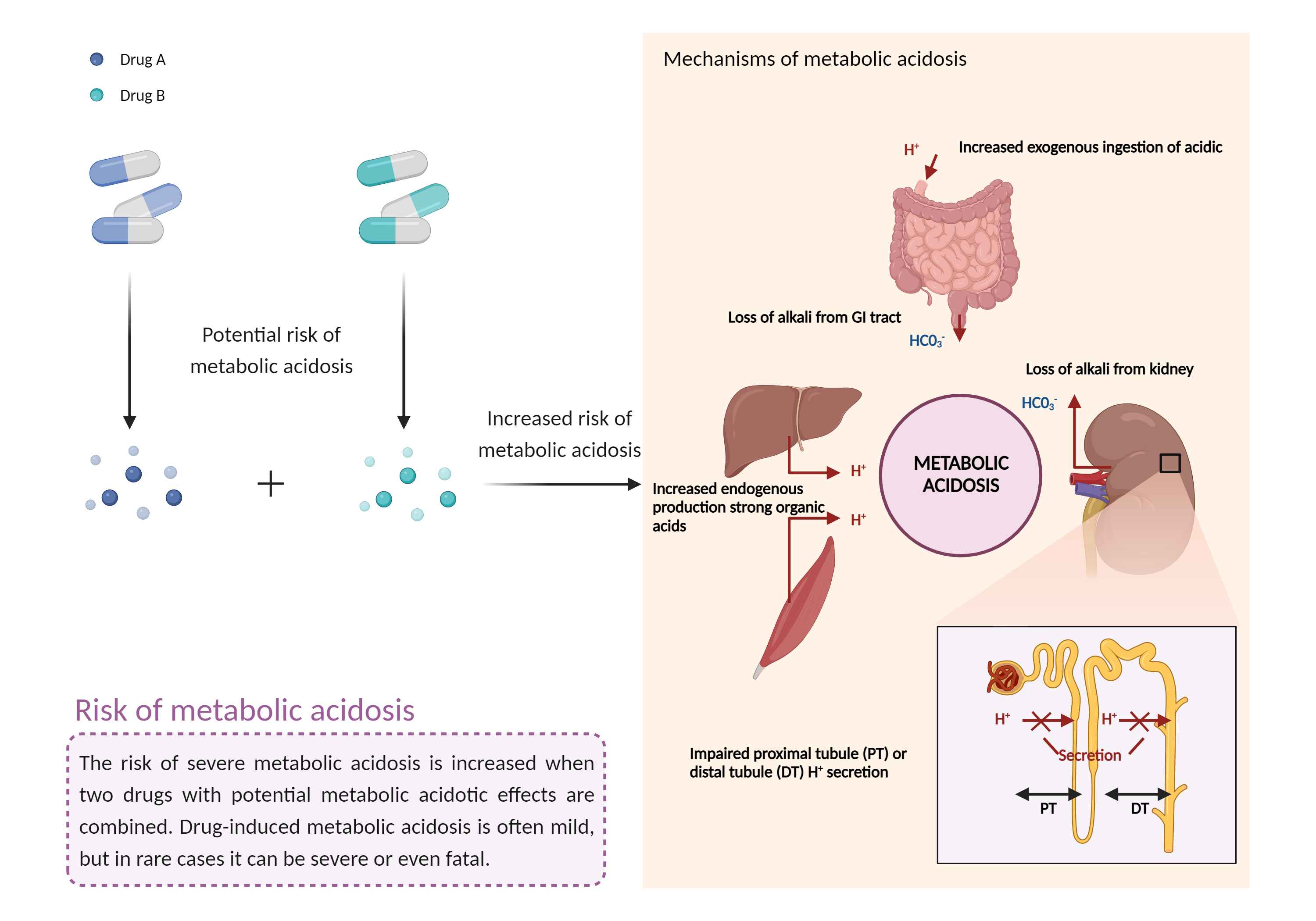
The use of more than one carbonic anhydrase inhibitor at a time is generally not recommended. Patients receiving treatment with a carbonic anhydrase inhibitor should have baseline and periodic measurements of serum bicarbonate. If metabolic acidosis develops and persists, consideration should be given to reducing the dosage or discontinuing treatment. Manifestations of acute or chronic metabolic acidosis may include hyperventilation, nonspecific symptoms such as fatigue and anorexia, or more severe sequelae like cardiac arrhythmias or stupor. If left untreated, metabolic acidosis can also lead to nephrolithiasis or nephrocalcinosis, osteomalacia (or rickets in children), osteoporosis, and reduced growth rates in pediatric patients. Increased fluid intake is recommended during therapy with carbonic anhydrase inhibitors to increase urinary output, which lowers the concentration of substances involved in stone formation. Patients, particularly pediatric patients, should be monitored closely for evidence of decreased sweating and increased body temperature, especially in warm or hot weather. Proper hydration before and during vigorous activities or exposure to warm temperatures is recommended. Patients (or their guardians or caregivers) should contact their doctor immediately if they develop signs or symptoms of kidney stones such as sudden back pain, abdominal pain and/or blood in the urine, of if they are not sweating as usual, with or without a fever. Carbonic anhydrase inhibitors should be used cautiously in the presence of predisposing factors to acidosis, such as renal impairment, severe respiratory disorders, status epilepticus, prolonged or severe diarrhea, a ketogenic diet (i.e., high protein/low carbohydrate), or surgery. |