Details of Drug-Drug Interaction
| Drug General Information (ID: DDISMPV0CF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Penicillamine | Drug Info | Ferrous sulfate anhydrous | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidotes | Iron Supplement | |||||||
| Structure | |||||||||
| Mechanism of Penicillamine-Ferrous sulfate anhydrous Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
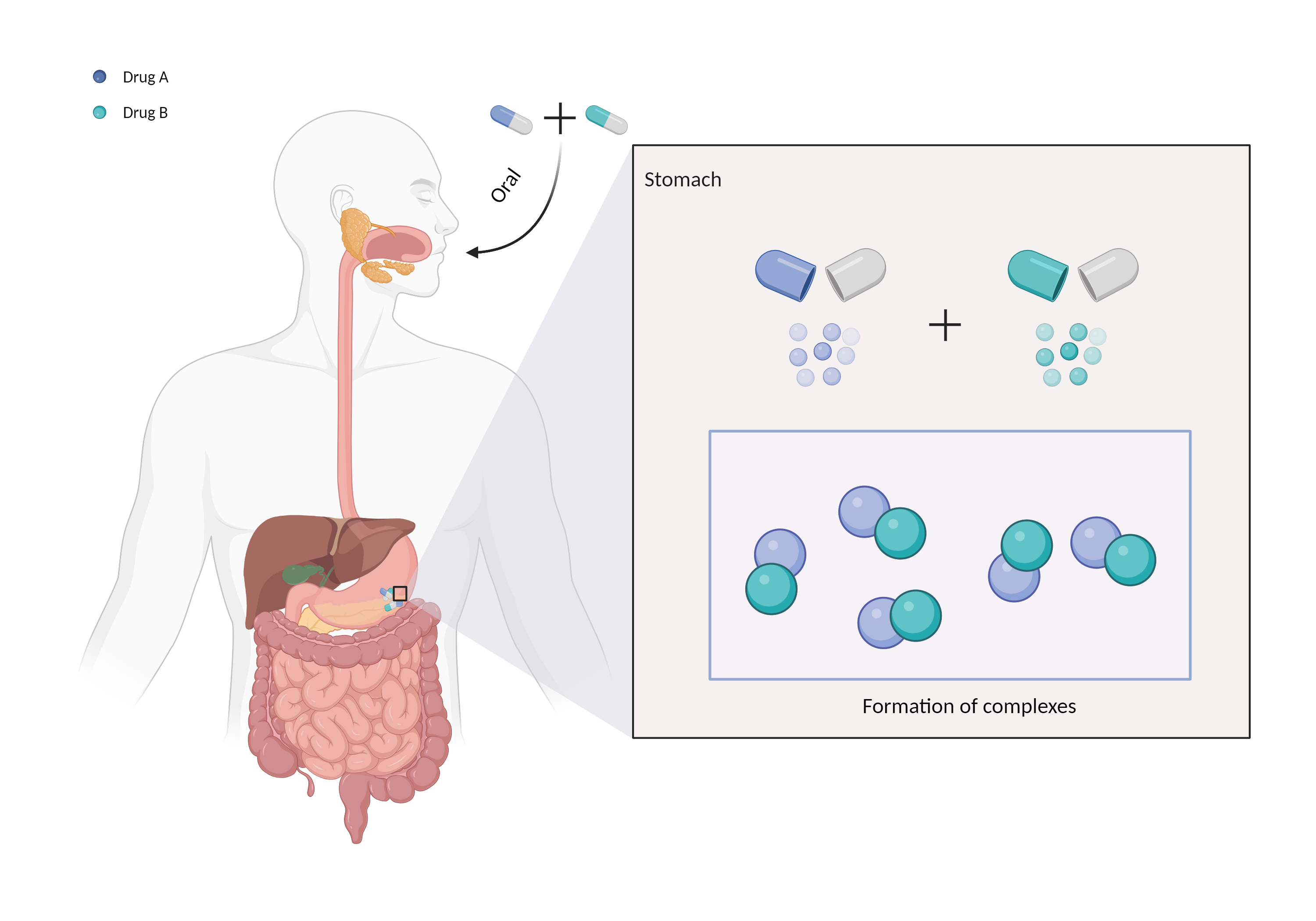
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Penicillamine | Ferrous sulfate anhydrous | |||||||
| Mechanism | Binds to polyvalent cations | Polyvalent cations | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Mineral supplements or other products containing polyvalent cations (e.g., antacids or preparations containing antacids such as didanosine buffered tablets or pediatric oral solution) should be administered at least two hours before or two hours after the penicillamine dose. In addition, pharmacologic response to penicillamine should be monitored more closely whenever these products are added to or withdrawn from therapy, and the penicillamine dosage adjusted as necessary. | ||||||||
