| Mechanism of Liothyronine-Iodide I-131 Interaction
(Severity Level: Moderate)
|
|
Interference of cell/tissue uptake
Click to Show/Hide Mechanism Graph
|
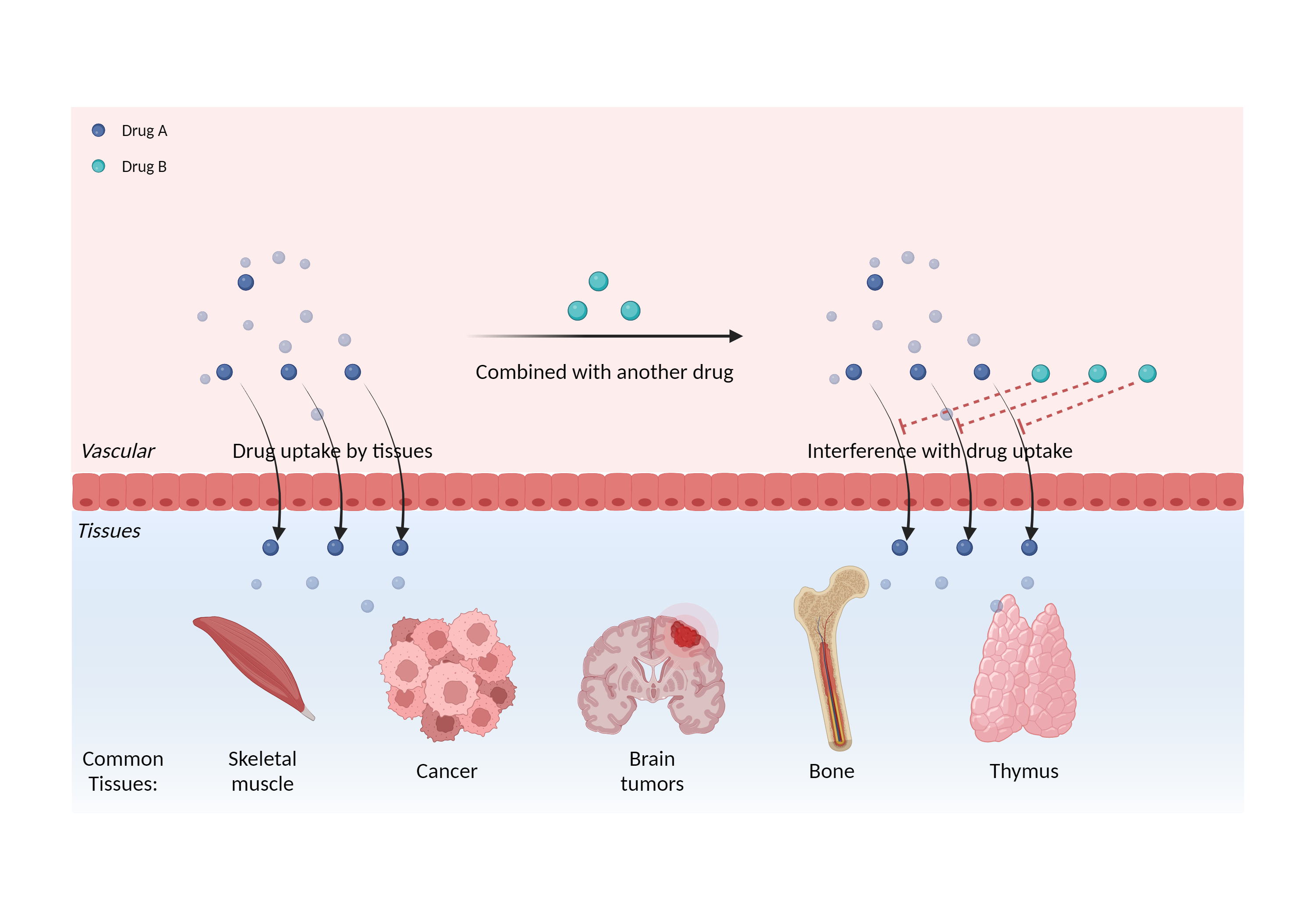 |
| Drug Name |
Liothyronine |
Iodide I-131 |
|
Mechanism
|
Interfere with the therapeutic effect and/or diagnostic result of radioiodides |
Radioiodide |
| Key Mechanism Factor 1 |
| Factor Name |
Pharmacokinetics |
| Factor Description |
Alters the distribution of the drug, leading to changes in the plasma concentration of the drug and affecting the pharmacological effects. |
| Mechanism Description |
- Interference of cell/tissue uptake of Iodide I-131 by Liothyronine
|

