Details of Drug-Drug Interaction
| Drug General Information (ID: DDIS5HA9RV) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Atropine (ophthalmic) | Drug Info | Physostigmine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiarrhythmic Agents | Ophthalmic Glaucoma Agents/Antidotes | |||||||
| Structure | |||||||||
| Mechanism of Atropine (ophthalmic)-Physostigmine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Antagonize the effect of cholinergic agents Click to Show/Hide Mechanism Graph | |||||||||
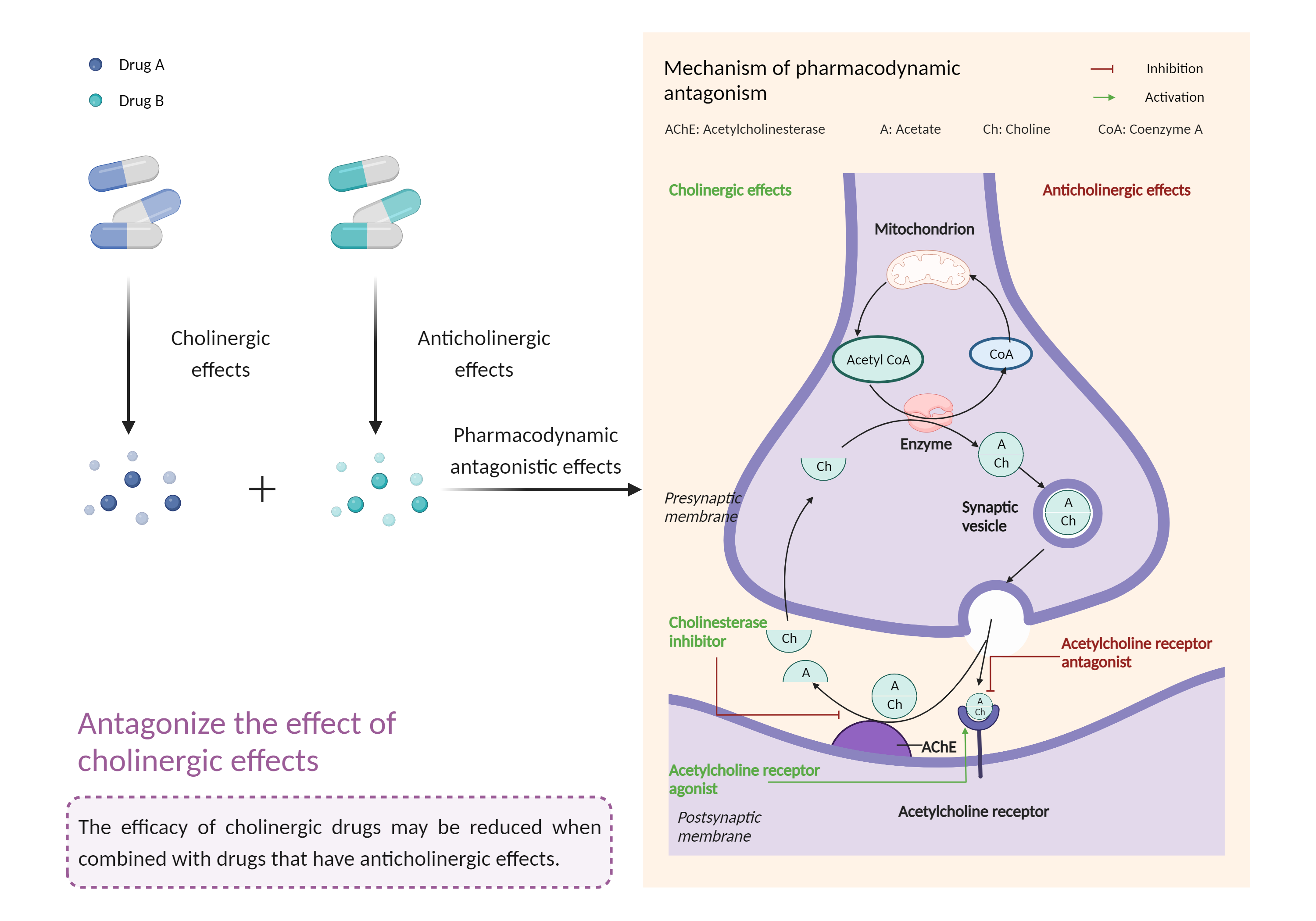 |
|||||||||
| Drug Name | Atropine (ophthalmic) | Physostigmine | |||||||
| Mechanism |
Anticholinergic effects Muscarinic acetylcholine receptor Antagonist |
Cholinergic effects Acetylcholinesterase Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Muscarinic acetylcholine receptor M | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
The muscarinic acetylcholine receptor mediates various cellular responses, including inhibition of adenylate cyclase, breakdown of phosphoinositides and modulation of potassium channels through the action of G proteins. Primary transducing effect is Pi turnover.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Acetylcholinesterase |
×
Structure
Sequence
MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPVSAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPNRELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSMNYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASVGMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTELVACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVGVVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPEDPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGYEIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQYVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKNQFDHYSKQDRCSDL
|
|||||||
| Gene Name | ACHE | ||||||||
| Uniprot ID | ACES_HUMAN | ||||||||
| KEGG Pathway | hsa:43 | ||||||||
| Protein Family | Type-B carboxylesterase/lipase family | ||||||||
| Protein Function |
Hydrolyzes rapidly the acetylcholine neurotransmitter released into the synaptic cleft allowing to terminate the signal transduction at the neuromuscular junction. Role in neuronal apoptosis.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Anticholinergic agents should generally be avoided in patients with Alzheimer's disease or other cognitive impairment, regardless of whether they are receiving an acetylcholinesterase inhibitor. For patients requiring treatment to counteract adverse effects of acetylcholinesterase inhibitor therapy (e.g., gastrointestinal intolerance, urinary problems), an agent without anticholinergic properties should be used whenever possible. Otherwise, a dosage reduction, slower titration, or even discontinuation of the acetylcholinesterase inhibitor should be considered. For patients who are already receiving an acetylcholinesterase inhibitor with anticholinergic agents, every attempt should be made to discontinue the latter or substitute them with less anticholinergic alternatives. Caution is required, however, since anticholinergic withdrawal may occur. Seizures have been reported following abrupt discontinuation of anticholinergics during acetylcholinesterase inhibitor therapy. | ||||||||
