Details of Drug-Drug Interaction
| Drug General Information (ID: DDIS2Y0LU9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Trandolapril | Drug Info | Meclofenamic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Analgesics | |||||||
| Structure | |||||||||
| Mechanism of Trandolapril-Meclofenamic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Antagonize the effect of antihypertensive agents Click to Show/Hide Mechanism Graph | |||||||||
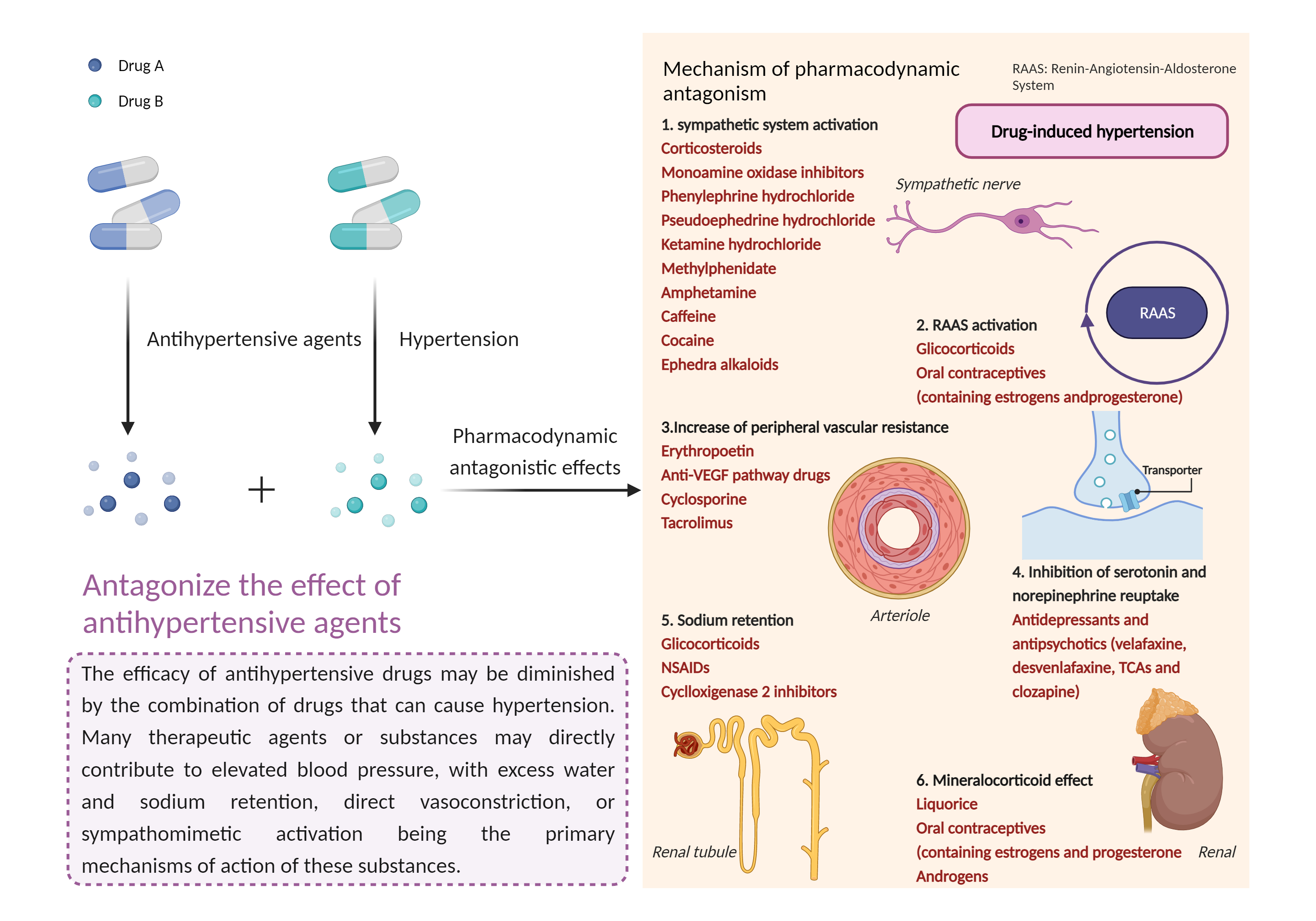 |
|||||||||
| Drug Name | Trandolapril | Meclofenamic acid | |||||||
| Mechanism |
Antihypertensive agent Angiotensin-converting enzyme Inhibitor |
Hypertensive effects Prostaglandin G/H synthase Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Angiotensin-converting enzyme |
×
Structure
Sequence
MGAASGRRGPGLLLPLPLLLLLPPQPALALDPGLQPGNFSADEAGAQLFAQSYNSSAEQVLFQSVAASWAHDTNITAENARRQEEAALLSQEFAEAWGQKAKELYEPIWQNFTDPQLRRIIGAVRTLGSANLPLAKRQQYNALLSNMSRIYSTAKVCLPNKTATCWSLDPDLTNILASSRSYAMLLFAWEGWHNAAGIPLKPLYEDFTALSNEAYKQDGFTDTGAYWRSWYNSPTFEDDLEHLYQQLEPLYLNLHAFVRRALHRRYGDRYINLRGPIPAHLLGDMWAQSWENIYDMVVPFPDKPNLDVTSTMLQQGWNATHMFRVAEEFFTSLELSPMPPEFWEGSMLEKPADGREVVCHASAWDFYNRKDFRIKQCTRVTMDQLSTVHHEMGHIQYYLQYKDLPVSLRRGANPGFHEAIGDVLALSVSTPEHLHKIGLLDRVTNDTESDINYLLKMALEKIAFLPFGYLVDQWRWGVFSGRTPPSRYNFDWWYLRTKYQGICPPVTRNETHFDAGAKFHVPNVTPYIRYFVSFVLQFQFHEALCKEAGYEGPLHQCDIYRSTKAGAKLRKVLQAGSSRPWQEVLKDMVGLDALDAQPLLKYFQPVTQWLQEQNQQNGEVLGWPEYQWHPPLPDNYPEGIDLVTDEAEASKFVEEYDRTSQVVWNEYAEANWNYNTNITTETSKILLQKNMQIANHTLKYGTQARKFDVNQLQNTTIKRIIKKVQDLERAALPAQELEEYNKILLDMETTYSVATVCHPNGSCLQLEPDLTNVMATSRKYEDLLWAWEGWRDKAGRAILQFYPKYVELINQAARLNGYVDAGDSWRSMYETPSLEQDLERLFQELQPLYLNLHAYVRRALHRHYGAQHINLEGPIPAHLLGNMWAQTWSNIYDLVVPFPSAPSMDTTEAMLKQGWTPRRMFKEADDFFTSLGLLPVPPEFWNKSMLEKPTDGREVVCHASAWDFYNGKDFRIKQCTTVNLEDLVVAHHEMGHIQYFMQYKDLPVALREGANPGFHEAIGDVLALSVSTPKHLHSLNLLSSEGGSDEHDINFLMKMALDKIAFIPFSYLVDQWRWRVFDGSITKENYNQEWWSLRLKYQGLCPPVPRTQGDFDPGAKFHIPSSVPYIRYFVSFIIQFQFHEALCQAAGHTGPLHKCDIYQSKEAGQRLATAMKLGFSRPWPEAMQLITGQPNMSASAMLSYFKPLLDWLRTENELHGEKLGWPQYNWTPNSARSEGPLPDSGRVSFLGLDLDAQQARVGQWLLLFLGIALLVATLGLSQRLFSIRHRSLHRHSHGPQFGSEVELRHS
|
|||||||
| Gene Name | ACE | ||||||||
| Uniprot ID | ACE_HUMAN | ||||||||
| KEGG Pathway | hsa:1636 | ||||||||
| Protein Family | Peptidase M2 family | ||||||||
| Protein Function |
Converts angiotensin I to angiotensin II by release of the terminal His-Leu, this results in an increase of the vasoconstrictor activity of angiotensin. Also able to inactivate bradykinin, a potent vasodilator. Has also a glycosidase activity which releases GPI-anchored proteins from the membrane by cleaving the mannose linkage in the GPI moiety.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Prostaglandin G/H synthase 2 |
×
Structure
Sequence
MLARALLLCAVLALSHTANPCCSHPCQNRGVCMSVGFDQYKCDCTRTGFYGENCSTPEFLTRIKLFLKPTPNTVHYILTHFKGFWNVVNNIPFLRNAIMSYVLTSRSHLIDSPPTYNADYGYKSWEAFSNLSYYTRALPPVPDDCPTPLGVKGKKQLPDSNEIVEKLLLRRKFIPDPQGSNMMFAFFAQHFTHQFFKTDHKRGPAFTNGLGHGVDLNHIYGETLARQRKLRLFKDGKMKYQIIDGEMYPPTVKDTQAEMIYPPQVPEHLRFAVGQEVFGLVPGLMMYATIWLREHNRVCDVLKQEHPEWGDEQLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNKQFQYQNRIAAEFNTLYHWHPLLPDTFQIHDQKYNYQQFIYNNSILLEHGITQFVESFTRQIAGRVAGGRNVPPAVQKVSQASIDQSRQMKYQSFNEYRKRFMLKPYESFEELTGEKEMSAELEALYGDIDAVELYPALLVEKPRPDAIFGETMVEVGAPFSLKGLMGNVICSPAYWKPSTFGGEVGFQIINTASIQSLICNNVKGCPFTSFSVPDPELIKTVTINASSSRSGLDDINPTVLLKERSTEL
|
|||||||
| Gene Name | PTGS2 | ||||||||
| Uniprot ID | PGH2_HUMAN | ||||||||
| KEGG Pathway | hsa:5743 | ||||||||
| Protein Family | Prostaglandin G/H synthase family | ||||||||
| Protein Function |
Dual cyclooxygenase and peroxidase in the biosynthesis pathway of prostanoids, a class of C20 oxylipins mainly derived from arachidonate, with a particular role in the inflammatory response (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593, PubMed:11939906, PubMed:19540099). The cyclooxygenase activity oxygenates arachidonate (AA, C20:4(n-6)) to the hydroperoxy endoperoxide prostaglandin G2 (PGG2), and the peroxidase activity reduces PGG2 to the hydroxy endoperoxide PGH2, the precursor of all 2-series prostaglandins and thromboxanes (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593). This complex transformation is initiated by abstraction of hydrogen at carbon 13 (with S-stereochemistry), followed by insertion of molecular O2 to form the endoperoxide bridge between carbon 9 and 11 that defines prostaglandins. The insertion of a second molecule of O2 (bis-oxygenase activity) yields a hydroperoxy group in PGG2 that is then reduced to PGH2 by two electrons (PubMed:7947975, PubMed:7592599, PubMed:9261177, PubMed:16373578, PubMed:22942274, PubMed:26859324, PubMed:27226593). Similarly catalyzes successive cyclooxygenation and peroxidation of dihomo-gamma-linoleate (DGLA, C20:3(n-6)) and eicosapentaenoate (EPA, C20:5(n-3)) to corresponding PGH1 and PGH3, the precursors of 1- and 3-series prostaglandins (PubMed:11939906, PubMed:19540099). In an alternative pathway of prostanoid biosynthesis, converts 2-arachidonoyl lysophopholipids to prostanoid lysophopholipids, which are then hydrolyzed by intracellular phospholipases to release free prostanoids (PubMed:27642067). Metabolizes 2-arachidonoyl glycerol yielding the glyceryl ester of PGH2, a process that can contribute to pain response (PubMed:22942274). Generates lipid mediators from n-3 and n-6 polyunsaturated fatty acids (PUFAs) via a lipoxygenase-type mechanism. Oxygenates PUFAs to hydroperoxy compounds and then reduces them to corresponding alcohols (PubMed:11034610, PubMed:11192938, PubMed:9048568, PubMed:9261177). Plays a role in the generation of resolution phase interaction products (resolvins) during both sterile and infectious inflammation (PubMed:12391014). Metabolizes docosahexaenoate (DHA, C22:6(n-3)) to 17R-HDHA, a precursor of the D-series resolvins (RvDs) (PubMed:12391014). As a component of the biosynthetic pathway of E-series resolvins (RvEs), converts eicosapentaenoate (EPA, C20:5(n-3)) primarily to 18S-HEPE that is further metabolized by ALOX5 and LTA4H to generate 18S-RvE1 and 18S-RvE2 (PubMed:21206090). In vascular endothelial cells, converts docosapentaenoate (DPA, C22:5(n-3)) to 13R-HDPA, a precursor for 13-series resolvins (RvTs) shown to activate macrophage phagocytosis during bacterial infection (PubMed:26236990). In activated leukocytes, contributes to oxygenation of hydroxyeicosatetraenoates (HETE) to diHETES (5,15-diHETE and 5,11-diHETE) (PubMed:22068350, PubMed:26282205). During neuroinflammation, plays a role in neuronal secretion of specialized preresolving mediators (SPMs) 15R-lipoxin A4 that regulates phagocytic microglia (By similarity).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients receiving ACE inhibitors who require prolonged (greater than 1 week) concomitant therapy with an NSAID should have blood pressure monitored more closely following initiation, discontinuation, or change of dosage of the NSAID. Renal function should also be evaluated periodically during prolonged coadministration. The interaction is not expected to occur with low doses (e.g., low-dose aspirin) or intermittent short-term administration of NSAIDs. | ||||||||
