Details of Drug-Drug Interaction
| Drug General Information (ID: DDIRU6MGJF) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Acetaminophen | Drug Info | Ceritinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Analgesics | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Acetaminophen-Ceritinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
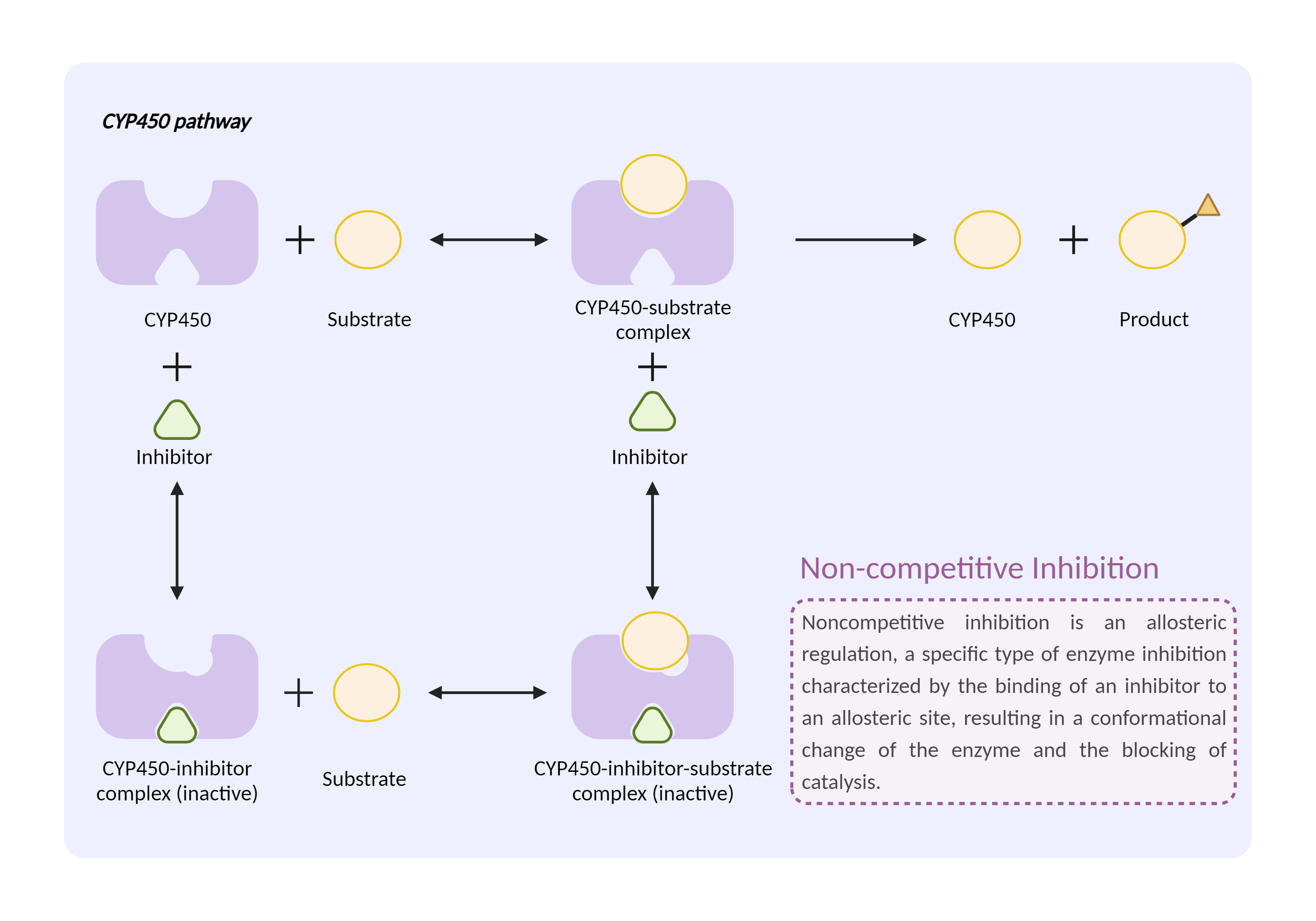 |
|||||||||
| Drug Name | Acetaminophen | Ceritinib | |||||||
| Mechanism 1 | CYP450 2A6 substrate | CYP450 2A6 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2A6 |
×
Structure
Sequence
MLASGMLLVALLVCLTVMVLMSVWQQRKSKGKLPPGPTPLPFIGNYLQLNTEQMYNSLMKISERYGPVFTIHLGPRRVVVLCGHDAVREALVDQAEEFSGRGEQATFDWVFKGYGVVFSNGERAKQLRRFSIATLRDFGVGKRGIEERIQEEAGFLIDALRGTGGANIDPTFFLSRTVSNVISSIVFGDRFDYKDKEFLSLLRMMLGIFQFTSTSTGQLYEMFSSVMKHLPGPQQQAFQLLQGLEDFIAKKVEHNQRTLDPNSPRDFIDSFLIRMQEEEKNPNTEFYLKNLVMTTLNLFIGGTETVSTTLRYGFLLLMKHPEVEAKVHEEIDRVIGKNRQPKFEDRAKMPYMEAVIHEIQRFGDVIPMSLARRVKKDTKFRDFFLPKGTEVYPMLGSVLRDPSFFSNPQDFNPQHFLNEKGQFKKSDAFVPFSIGKRNCFGEGLARMELFLFFTTVMQNFRLKSSQSPKDIDVSPKHVGFATIPRNYTMSFLPR
|
|||||||
| Gene Name | CYP2A6 | ||||||||
| Uniprot ID | CP2A6_HUMAN | ||||||||
| KEGG Pathway | hsa:1548 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
Exhibits a high coumarin 7-hydroxylase activity. Can act in the hydroxylation of the anti-cancer drugs cyclophosphamide and ifosphamide. Competent in the metabolic activation of aflatoxin B1. Constitutes the major nicotine C-oxidase. Acts as a 1,4-cineole 2-exo-monooxygenase. Possesses low phenacetin O-deethylation activity.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | CYP450 2E1 substrate | CYP450 2E1 inhibitor | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Cytochrome P450 2E1 |
×
Structure
Sequence
MSALGVTVALLVWAAFLLLVSMWRQVHSSWNLPPGPFPLPIIGNLFQLELKNIPKSFTRLAQRFGPVFTLYVGSQRMVVMHGYKAVKEALLDYKDEFSGRGDLPAFHAHRDRGIIFNNGPTWKDIRRFSLTTLRNYGMGKQGNESRIQREAHFLLEALRKTQGQPFDPTFLIGCAPCNVIADILFRKHFDYNDEKFLRLMYLFNENFHLLSTPWLQLYNNFPSFLHYLPGSHRKVIKNVAEVKEYVSERVKEHHQSLDPNCPRDLTDCLLVEMEKEKHSAERLYTMDGITVTVADLFFAGTETTSTTLRYGLLILMKYPEIEEKLHEEIDRVIGPSRIPAIKDRQEMPYMDAVVHEIQRFITLVPSNLPHEATRDTIFRGYLIPKGTVVVPTLDSVLYDNQEFPDPEKFKPEHFLNENGKFKYSDYFKPFSTGKRVCAGEGLARMELFLLLCAILQHFNLKPLVDPKDIDLSPIHIGFGCIPPRYKLCVIPRS
|
|||||||
| Gene Name | CYP2E1 | ||||||||
| Uniprot ID | CP2E1_HUMAN | ||||||||
| KEGG Pathway | hsa:1571 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of fatty acids (PubMed:10553002, PubMed:18577768). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:10553002, PubMed:18577768). Catalyzes the hydroxylation of carbon-hydrogen bonds. Hydroxylates fatty acids specifically at the omega-1 position displaying the highest catalytic activity for saturated fatty acids (PubMed:10553002, PubMed:18577768). May be involved in the oxidative metabolism of xenobiotics (Probable).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if ceritinib is used concomitantly with drugs that are substrates of CYP450 2A6 and/or 2E1, particularly those with a narrow therapeutic range. Dosage adjustments as well as clinical and laboratory monitoring should be considered whenever ceritinib is added to or withdrawn from therapy with these drugs. Patients should be monitored for the development of adverse effects. | ||||||||
