Details of Drug-Drug Interaction
| Drug General Information (ID: DDIR578Q4B) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Valproic acid | Drug Info | Cholestyramine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticonvulsants | Bile Acid Sequestrants | |||||||
| Structure | |||||||||
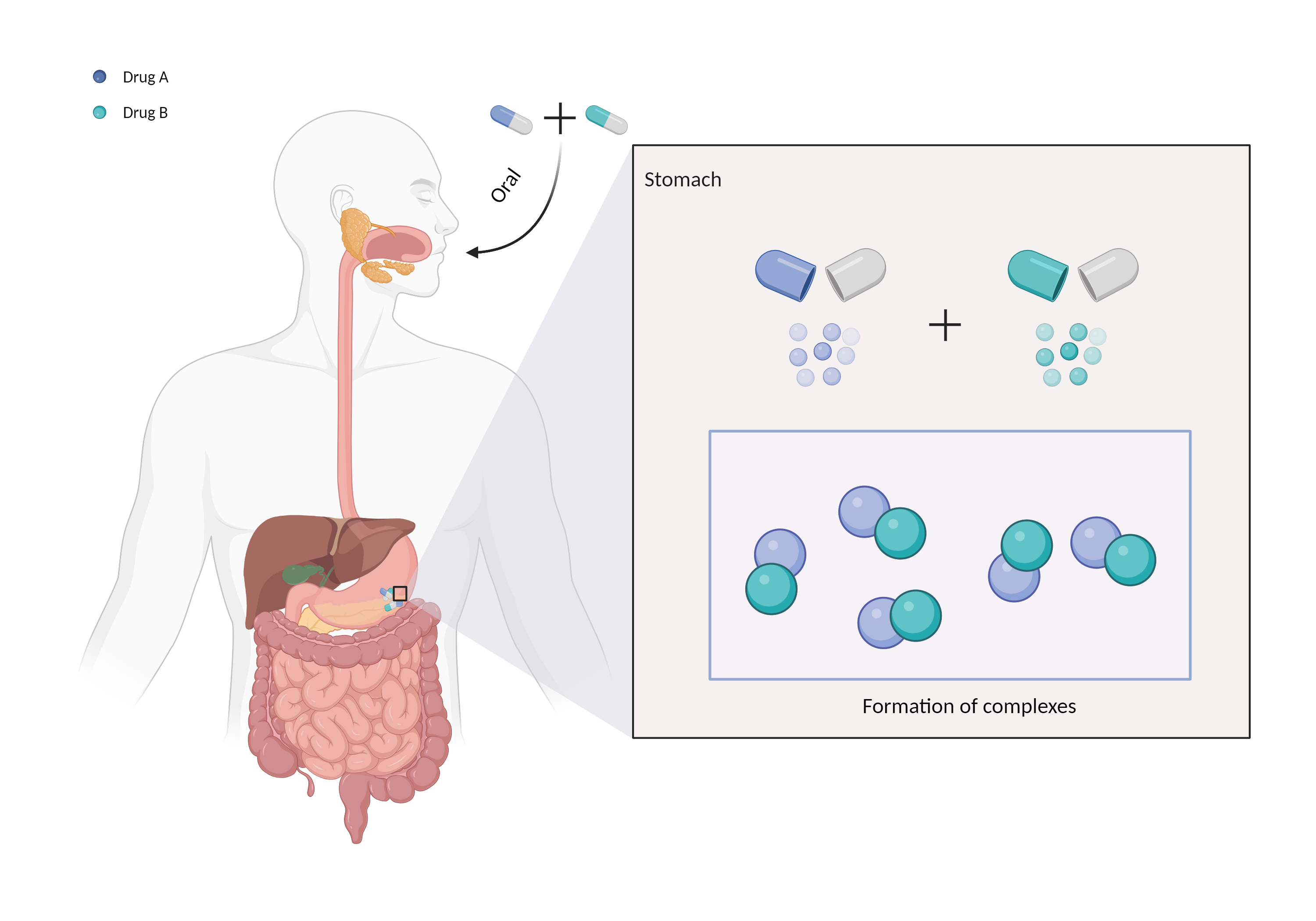
| Mechanism of Valproic acid-Cholestyramine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Valproic acid | Cholestyramine | |||||||
| Mechanism | Binds to anion exchange resin/polymer | Anion exchange resin/polymer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | To minimize potential for interaction, valproic acid and cholestyramine should be administered at least 3 hours apart. Patients should be monitored for clinical and laboratory evidence of altered valproate efficacy, and the dosage adjusted if necessary. It is not known whether these precautions are applicable to extended-release valproic acid or divalproex formulations. | ||||||||
