Details of Drug-Drug Interaction
| Drug General Information (ID: DDIR14ALC3) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Labetalol | Drug Info | Methyldopa | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Antihypertensive Agents | |||||||
| Structure | |||||||||
| Mechanism of Labetalol-Methyldopa Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
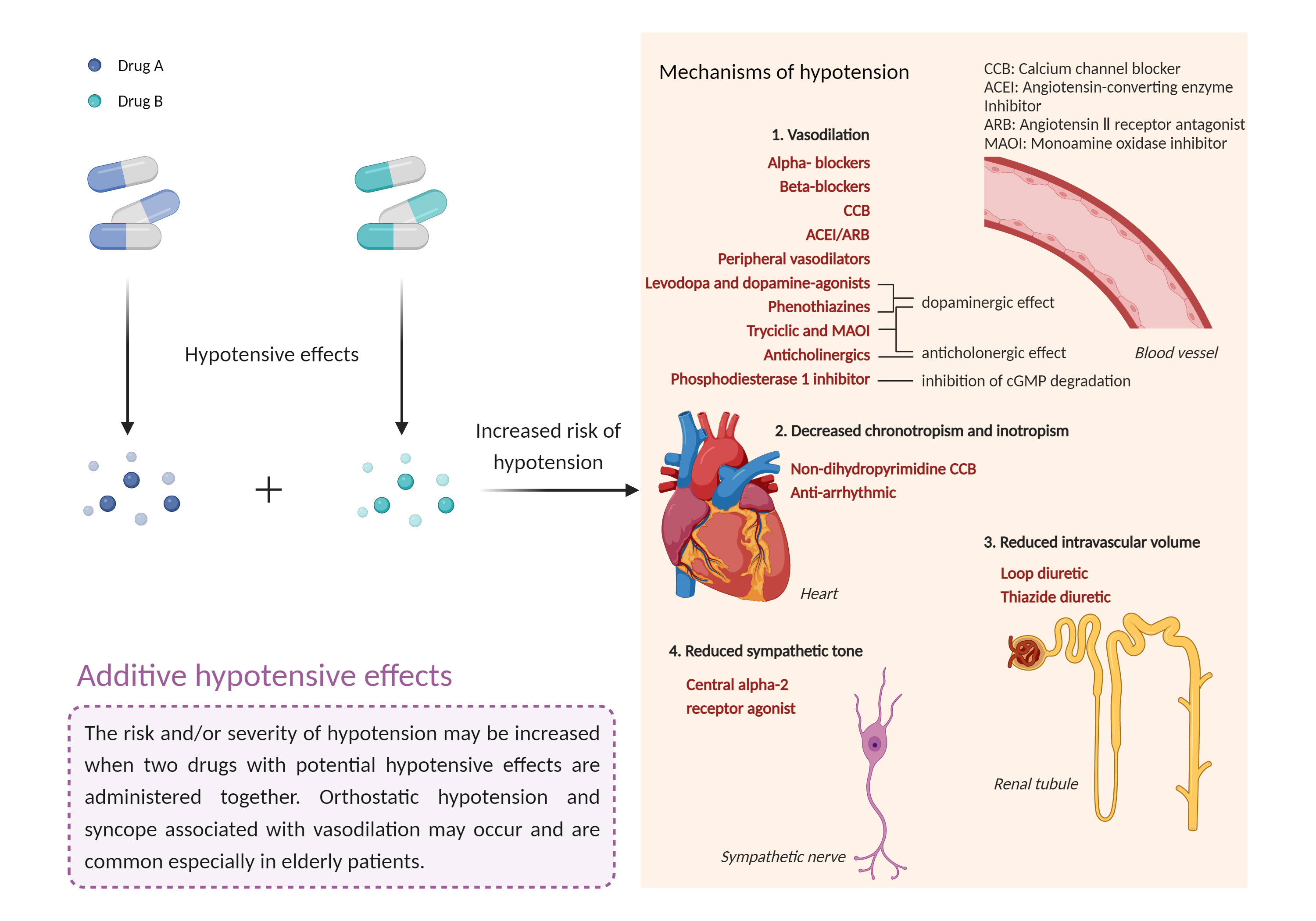 |
|||||||||
| Drug Name | Labetalol | Methyldopa | |||||||
| Mechanism |
Antihypertensive agent Alpha-1 adrenergic receptor Antagonist |
Antihypertensive agent Alpha-2 adrenergic receptor Agonist |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Adrenergic receptor alpha-1 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
This alpha-adrenergic receptor mediates its action by association with G proteins that activate a phosphatidylinositol-calcium second messenger system. Its effect is mediated by G(q) and G(11) proteins. Nuclear ADRA1A-ADRA1B heterooligomers regulate phenylephrine(PE)-stimulated ERK signaling in cardiac myocytes.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Adrenergic receptor alpha-2 | Structure Sequence | |||||||
| Protein Family | G-protein coupled receptor 1 family | ||||||||
| Protein Function |
Alpha-2 adrenergic receptors mediate the catecholamine-induced inhibition of adenylate cyclase through the action of G proteins. The rank order of potency for agonists of this receptor is oxymetazoline > clonidine > epinephrine > norepinephrine > phenylephrine > dopamine > p-synephrine > p-tyramine > serotonin = p-octopamine. For antagonists, the rank order is yohimbine > phentolamine = mianserine > chlorpromazine = spiperone = prazosin > propanolol > alprenolol = pindolol.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close monitoring of blood pressure is recommended during concomitant use, and if methyldopa or the beta blocker are withdrawn from therapy. Methyldopa manufacturers recommend adjusting the beta blocker dose if methyldopa is added to therapy, and not exceeding a methyldopa dose of 500 mg/day when it is first added to therapy. Patients should be instructed to notify their doctor if they have a reduced heart rate, dizziness, fainting or headaches, chest pain or vision problems. | ||||||||
