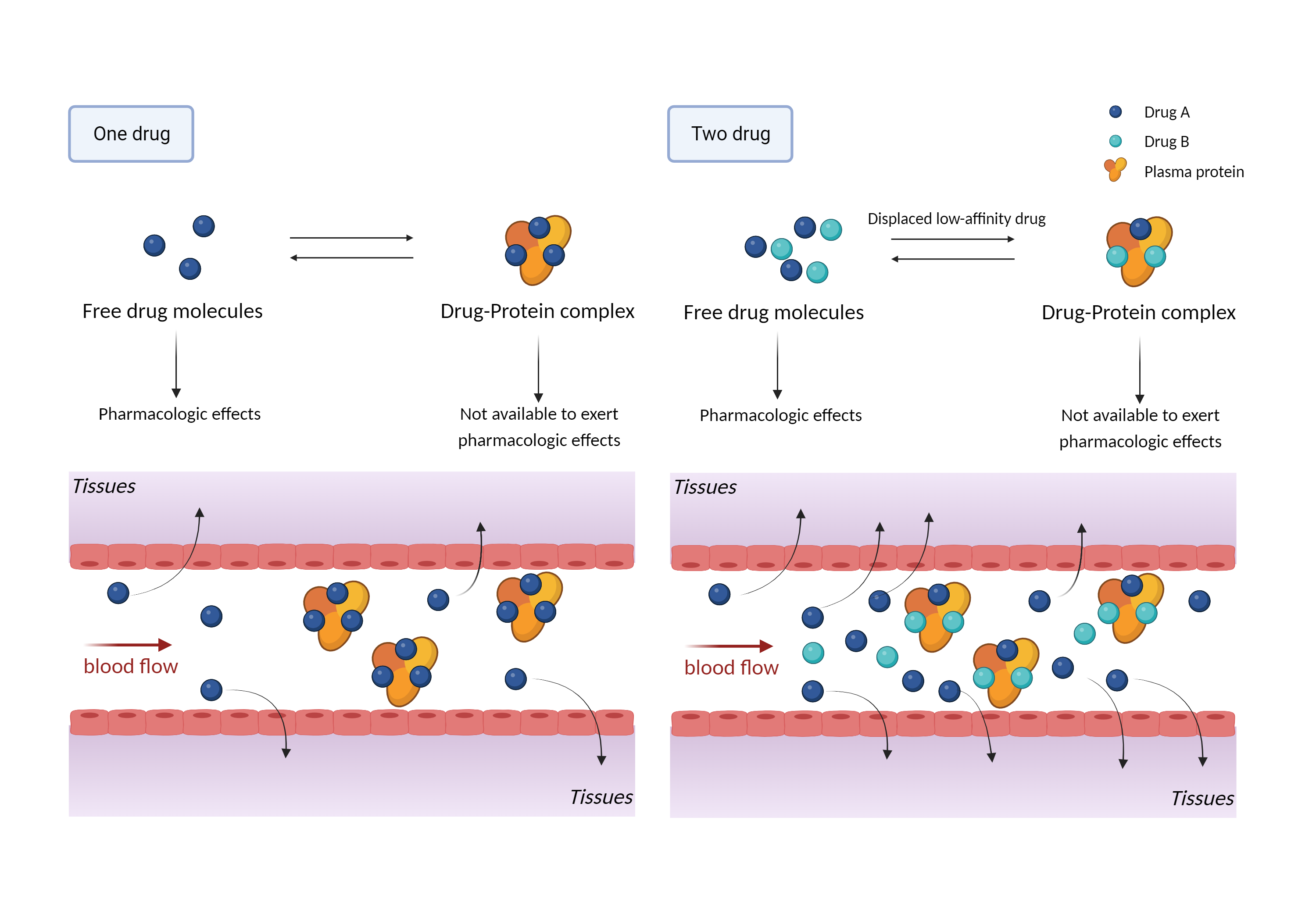
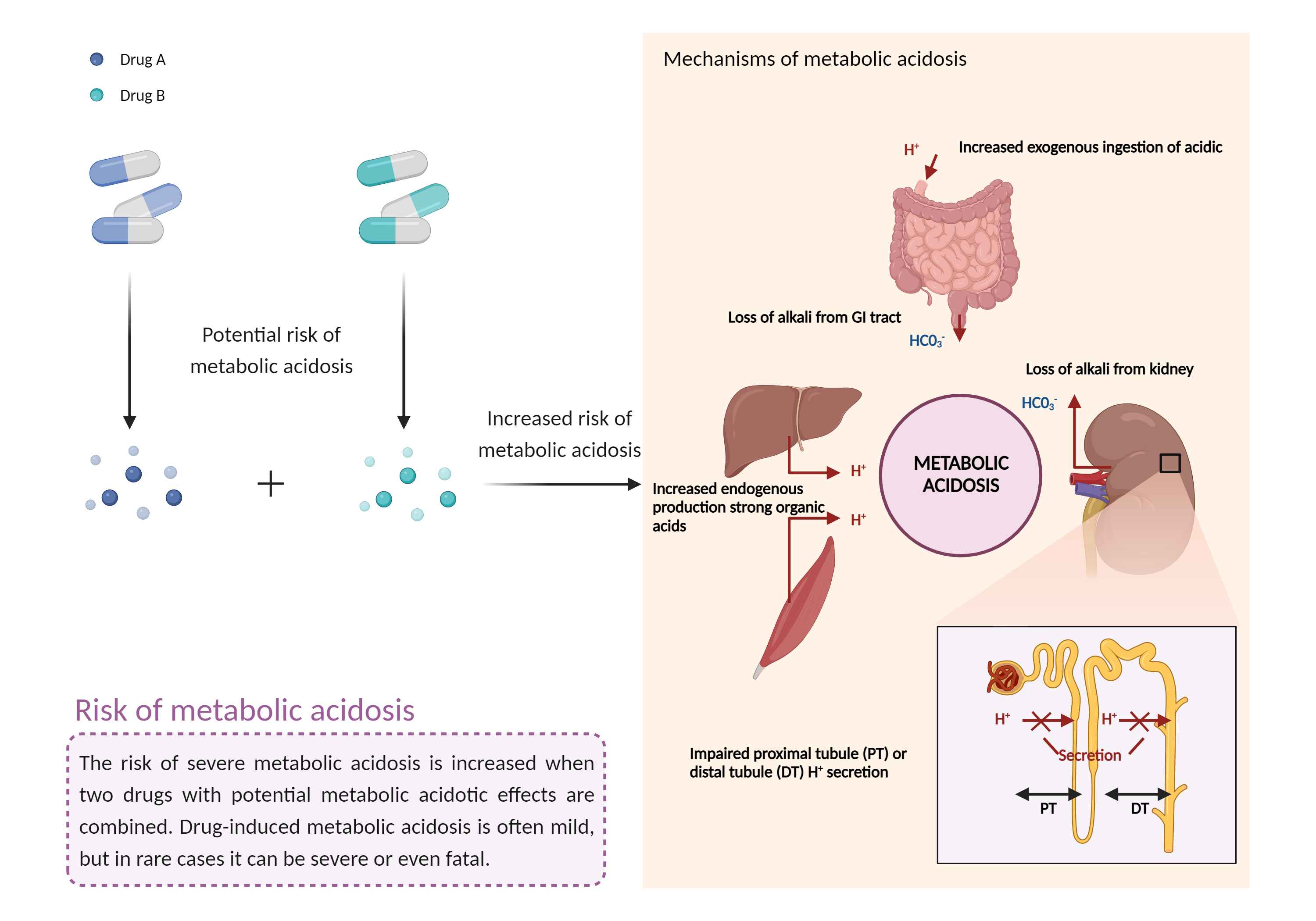
| References |
| 1 |
Anderson CJ, Kaufman PL, Sturm RJ "Toxicity of combined therapy with carbonic anhydrase inhibitors and aspirin." Am J Ophthalmol 86 (1978): 516-9. [PMID: 707596]
|
| 2 |
Canadian Pharmacists Association.
|
| 3 |
Cerner Multum, Inc. "Australian Product Information.".
|
| 4 |
Cerner Multum, Inc. "UK Summary of Product Characteristics.".
|
| 5 |
Cowan RA, Hartnell GG, Lowdell CP, Baird IM, Leak AM "Metabolic acidosis induced by carbonic anhydrase inhibitors and salicylates in patients with normal renal function." Br Med J (Clin Res Ed) 289 (1984): 347-8. [PMID: 6432091]
|
| 6 |
Favre L, Vallotton MB "Relationship of renal prostaglandins to three diuretics." Prostaglandins Leukot Med 14 (1984): 313-9. [PMID: 6589647]
|
| 7 |
Product Information. Azopt (brinzolamide ophthalmic). Alcon Laboratories Inc, Fort Worth, TX.
|
| 8 |
Product Information. Diamox (acetazolamide). Lederle Laboratories, Wayne, NJ.
|
| 9 |
Product Information. Trusopt (dorzolamide ophthalmic). Merck & Co, Inc, West Point, PA.
|
| 10 |
Rousseau P, Fuentevilla-Clifton A "Acetazolamide and salicylate interaction in the elderly: a case report." J Am Geriatr Soc 41 (1993): 868-9. [PMID: 8340566]
|
| 11 |
Sweeney KR, Chapron DJ, Antal EJ, Kramer PA "Differential effects of flurbiprofen and aspirin on acetazolamide disposition in humans." Br J Clin Pharmacol 27 (1989): 866-9. [PMID: 2757898]
|
| 12 |
Sweeney KR, Chapron DJ, Brandt JL, Gomolin IH, Feig PU, Kramer PA "Toxic interaction between acetazolamide and salicylate: case reports and a pharmacokinetic explanation." Clin Pharmacol Ther 40 (1986): 518-24. [PMID: 3769383]
|
| 13 |
Sweeney KR, Chapron DJ, Kramer PA "Effect of salicylate on serum protein binding and red blood cell uptake of acetazolamide in vitro." J Pharm Sci 77 (1988): 751-6. [PMID: 3225768]
|