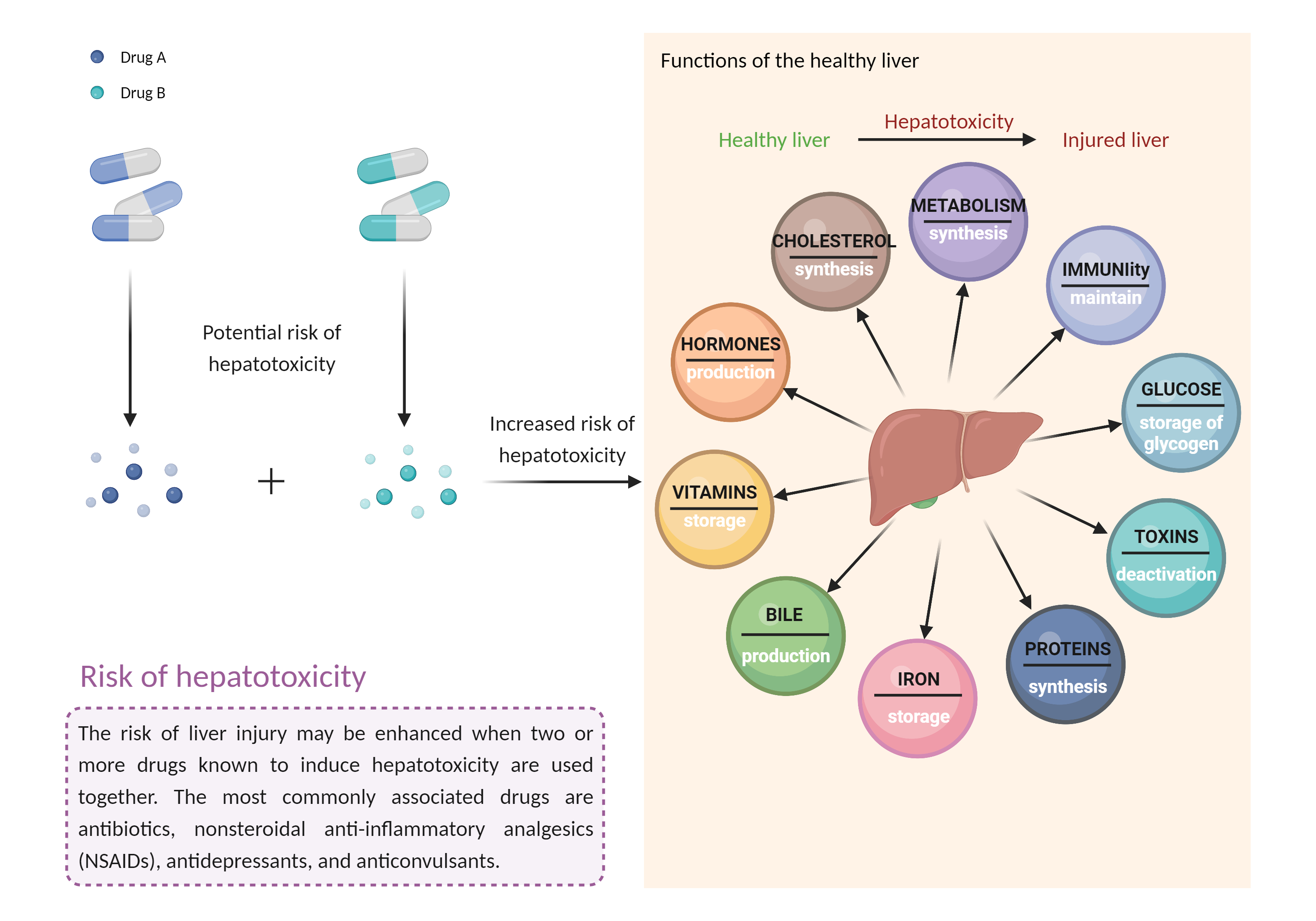
| Management |
Caution is advised if leflunomide or teriflunomide must be used in patients who are currently receiving or have recently received treatment with other hepatotoxic agents or are receiving drugs that are substrates of OATP 1B1/1B3 and/or BCRP, such as rosuvastatin. Liver enzymes and bilirubin should be measured prior to initiation of leflunomide/teriflunomide therapy and at least monthly for the first six months of treatment and every 6 to 8 weeks thereafter. Patients with preexisting liver disease or elevated baseline liver enzymes (i.e., ALT greater than two times ULN) should not receive leflunomide or teriflunomide. Patients who develop elevated serum ALT greater than three times ULN while receiving these medications should discontinue treatment and be given washout procedures with cholestyramine or activated charcoal to accelerate elimination of leflunomide's active metabolite from plasma, which otherwise may take up to two years. Follow-up monitoring should be conducted at least weekly until the ALT value is within normal range, and washout procedures repeated as necessary. All patients treated with leflunomide or teriflunomide should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. If concomitant administration is required, the dose of rosuvastatin should not exceed 10 mg once daily. Dosage adjustments as well as clinical and laboratory monitoring should be considered whenever leflunomide or teriflunomide is added to or withdrawn from therapy with rosuvastatin. |