Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQF07CVT) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Grepafloxacin | Drug Info | Methyl aminolevulinate (topical) | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antibiotics | Antineoplastics | |||||||
| Structure | |||||||||
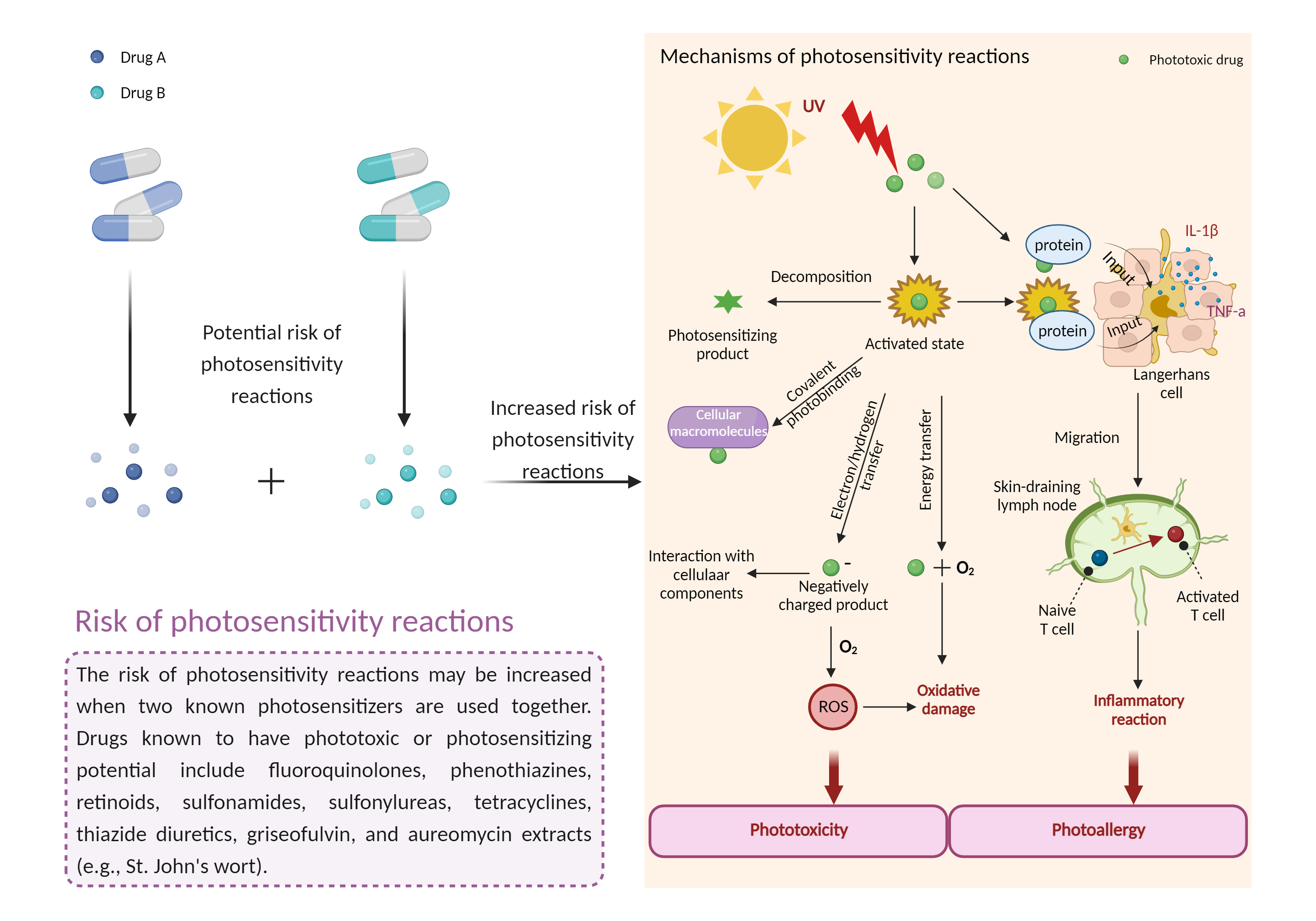
| Mechanism of Grepafloxacin-Methyl aminolevulinate (topical) Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of photosensitivity reactions Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Grepafloxacin | Methyl aminolevulinate (topical) | |||||||
| Mechanism | Photosensitivity reactions | Photosensitivity reactions | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Photosensitivity reactions | ||||||||
| Factor Description | Drug-induced photosensitivity reactions occur when certain photosensitizing drugs cause unexpected sunburn or dermatitis (dry, bumpy or blistering rash) on sun-exposed skin (face, neck, arms, backs of hands, and often lower legs and feet). The rash may or may not be itchy. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised and pharmacologic response to photodynamic therapy should be carefully monitored if concomitant use of other photosensitizing agents cannot be avoided. Patients should be advised to avoid exposure of treated areas to sunlight or bright indoor lights (e.g., examination lamps, operating room lamps, tanning beds, lights at close proximity) during the period between application of aminolevulinic acid or methyl aminolevulinate and photoactivation, and for 48 hours post-illumination. Concomitant use with other topical medicinal products should be avoided. | ||||||||
