Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQ7B0HIO) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Gemfibrozil | Drug Info | Enzalutamide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihyperammonia Agents | Antiandrogens | |||||||
| Structure | |||||||||
| Mechanism of Gemfibrozil-Enzalutamide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
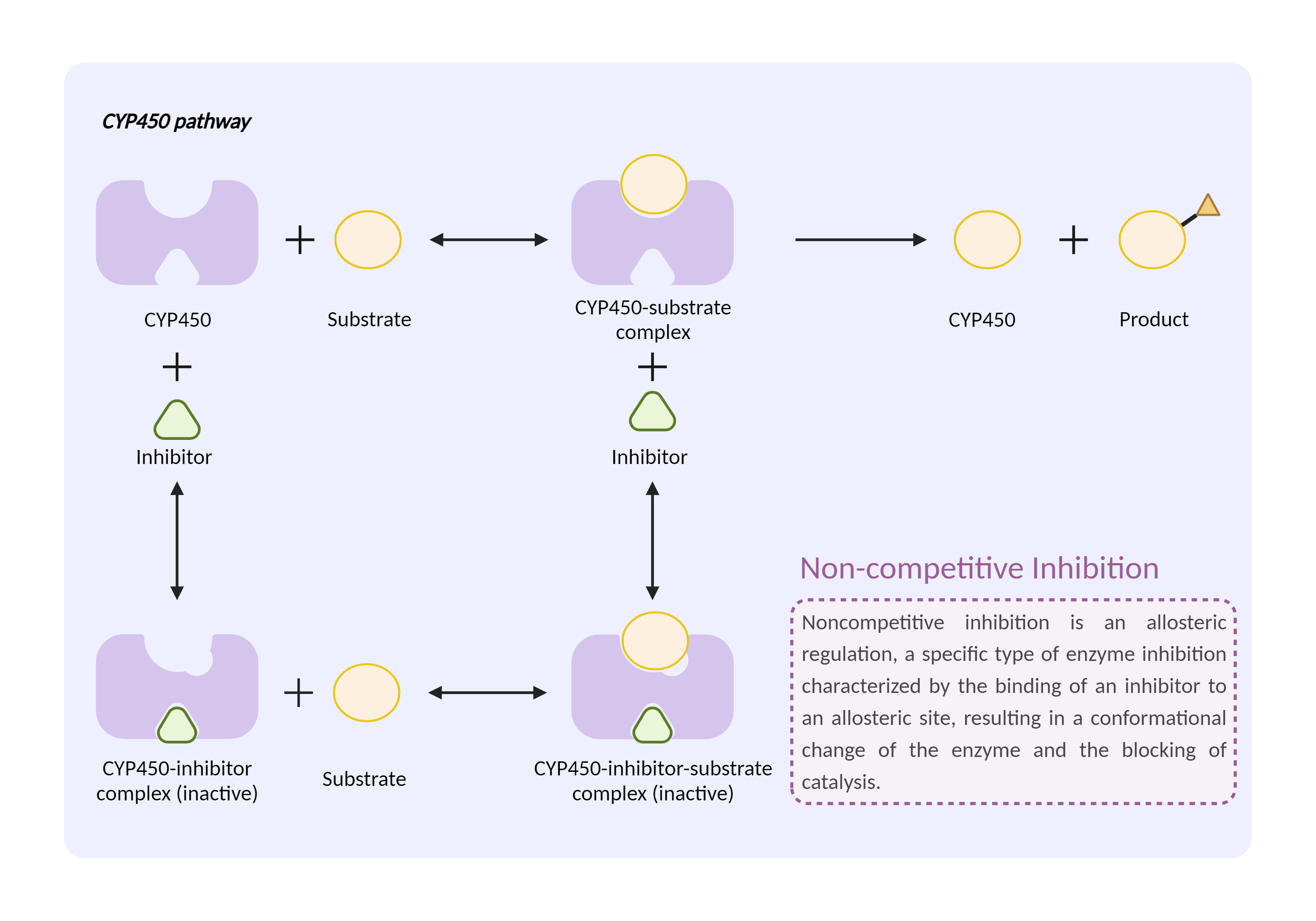 |
|||||||||
| Drug Name | Gemfibrozil | Enzalutamide | |||||||
| Mechanism | CYP450 2C8 inhibitor | CYP450 2C8 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C8 |
×
Structure
Sequence
MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKVYGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRWKEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICSVVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALTRSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTETTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSDLVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFKKSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLPPSYQICFIPV
|
|||||||
| Gene Name | CYP2C8 | ||||||||
| Uniprot ID | CP2C8_HUMAN | ||||||||
| KEGG Pathway | hsa:1558 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:11093772, PubMed:14559847, PubMed:15766564, PubMed:19965576). Primarily catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) with a preference for the last double bond (PubMed:7574697, PubMed:15766564, PubMed:19965576). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes all trans-retinoic acid toward its 4-hydroxylated form (PubMed:11093772). Displays 16-alpha hydroxylase activity toward estrogen steroid hormones, 17beta-estradiol (E2) and estrone (E1) (PubMed:14559847). Plays a role in the oxidative metabolism of xenobiotics. It is the principal enzyme responsible for the metabolism of the anti-cancer drug paclitaxel (taxol) (PubMed:26427316).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of enzalutamide in combination with potent CYP450 2C8 inhibitors such as gemfibrozil should generally be avoided. If concomitant use is required, a reduction of the enzalutamide dosage to 80 mg once daily is recommended. Following discontinuation of the potent CYP450 2C8 inhibitor, the enzalutamide dosage should be returned to that used prior to initiation of the inhibitor. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Xtandi (enzalutamide). Astellas Pharma US, Inc, Deerfield, IL. | ||||||||||||||||||
