Details of Drug-Drug Interaction
| Drug General Information (ID: DDIQ3KWUH9) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Chlorothiazide | Drug Info | Safinamide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Dopaminergic Antiparkinsonism Agents | |||||||
| Structure | |||||||||
| Mechanism of Chlorothiazide-Safinamide Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive hypotensive effects Click to Show/Hide Mechanism Graph | |||||||||
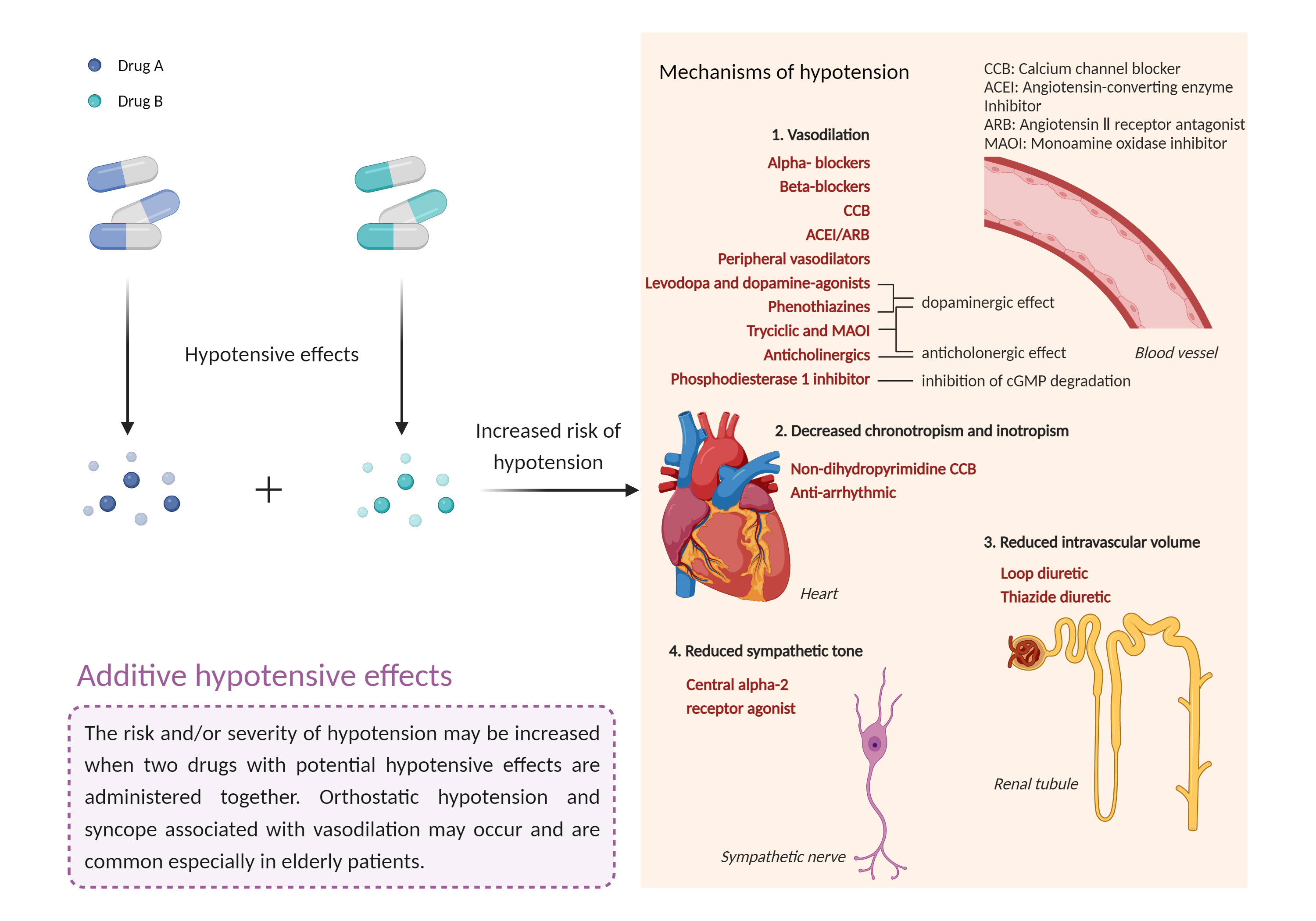 |
|||||||||
| Drug Name | Chlorothiazide | Safinamide | |||||||
| Mechanism |
Hypotensive effects Carbonic anhydrase II Inhibitor |
Hypotensive effects Monoamine oxidase-B selective Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Carbonic anhydrase II |
×
Structure
Sequence
MSHHWGYGKHNGPEHWHKDFPIAKGERQSPVDIDTHTAKYDPSLKPLSVSYDQATSLRILNNGHAFNVEFDDSQDKAVLKGGPLDGTYRLIQFHFHWGSLDGQGSEHTVDKKKYAAELHLVHWNTKYGDFGKAVQQPDGLAVLGIFLKVGSAKPGLQKVVDVLDSIKTKGKSADFTNFDPRGLLPESLDYWTYPGSLTTPPLLECVTWIVLKEPISVSSEQVLKFRKLNFNGEGEPEELMVDNWRPAQPLKNRQIKASFK
|
|||||||
| Gene Name | CA2 | ||||||||
| Uniprot ID | CAH2_HUMAN | ||||||||
| KEGG Pathway | hsa:760 | ||||||||
| Protein Family | Alpha-carbonic anhydrase family | ||||||||
| Protein Function |
Catalyzes the reversible hydration of carbon dioxide (PubMed:1909891, PubMed:1910042, PubMed:1336460, PubMed:8485129, PubMed:8399159, PubMed:8218160, PubMed:8262987, PubMed:8451242, PubMed:7901850, PubMed:7761440, PubMed:8639494, PubMed:9265618, PubMed:17330962, PubMed:9398308, PubMed:11327835, PubMed:12056894, PubMed:17346964, PubMed:12171926, PubMed:15453828, PubMed:16214338, PubMed:15865431, PubMed:16106378, PubMed:15300855, PubMed:15667203, PubMed:18942852, PubMed:11831900, PubMed:17251017). Can also hydrate cyanamide to urea (PubMed:10550681, PubMed:11015219). Stimulates the chloride-bicarbonate exchange activity of SLC26A6 (PubMed:15990874). Essential for bone resorption and osteoclast differentiation (PubMed:15300855). Involved in the regulation of fluid secretion into the anterior chamber of the eye. Contributes to intracellular pH regulation in the duodenal upper villous epithelium during proton-coupled peptide absorption.
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Monoamine oxidase type B |
×
Structure
Sequence
MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSYVGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWRTMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEVSALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQTRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVYYKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEERLKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDRIYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTTFLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|||||||
| Gene Name | MAOB | ||||||||
| Uniprot ID | AOFB_HUMAN | ||||||||
| KEGG Pathway | hsa:4129 | ||||||||
| Protein Family | Flavin monoamine oxidase family | ||||||||
| Protein Function |
Catalyzes the oxidative deamination of primary and some secondary amines such as neurotransmitters, and exogenous amines including the tertiary amine, neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), with concomitant reduction of oxygen to hydrogen peroxide and participates in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079). Preferentially degrades benzylamine and phenylethylamine (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised during coadministration of MAOIs and other medications with hypotensive effects, especially during the first few weeks of treatment. Close monitoring for development of hypotension is recommended. Ambulatory patients should be advised to avoid rising abruptly from a sitting or recumbent position and to notify their physician if they experience dizziness, lightheadedness, syncope, orthostasis, or tachycardia. | ||||||||
