Details of Drug-Drug Interaction
| Drug General Information (ID: DDIPVNSJXE) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Cyclophosphamide | Drug Info | Azathioprine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Immunosuppressive Agents | |||||||
| Structure | |||||||||
| Mechanism of Cyclophosphamide-Azathioprine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
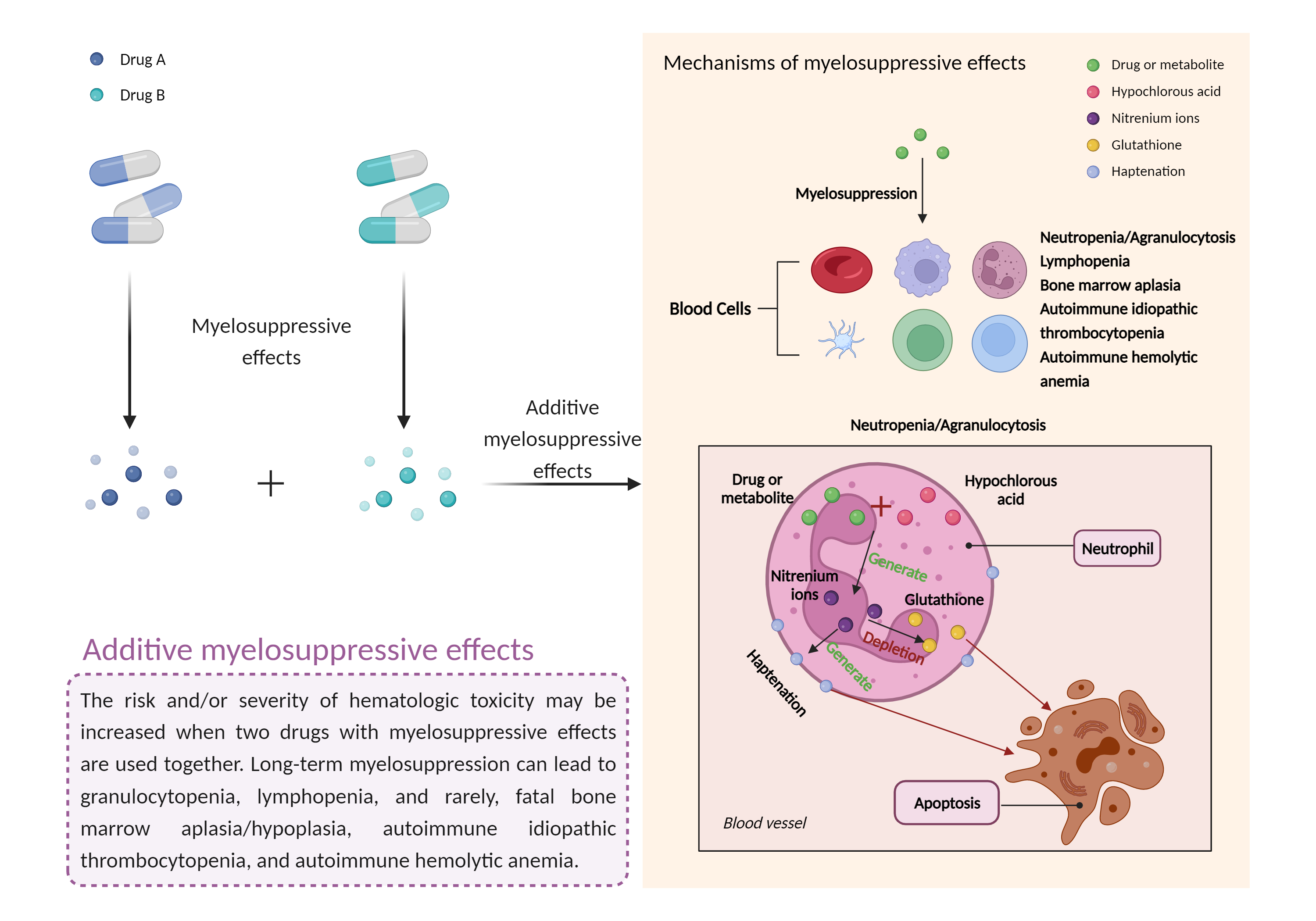
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cyclophosphamide | Azathioprine | |||||||
| Mechanism 1 | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
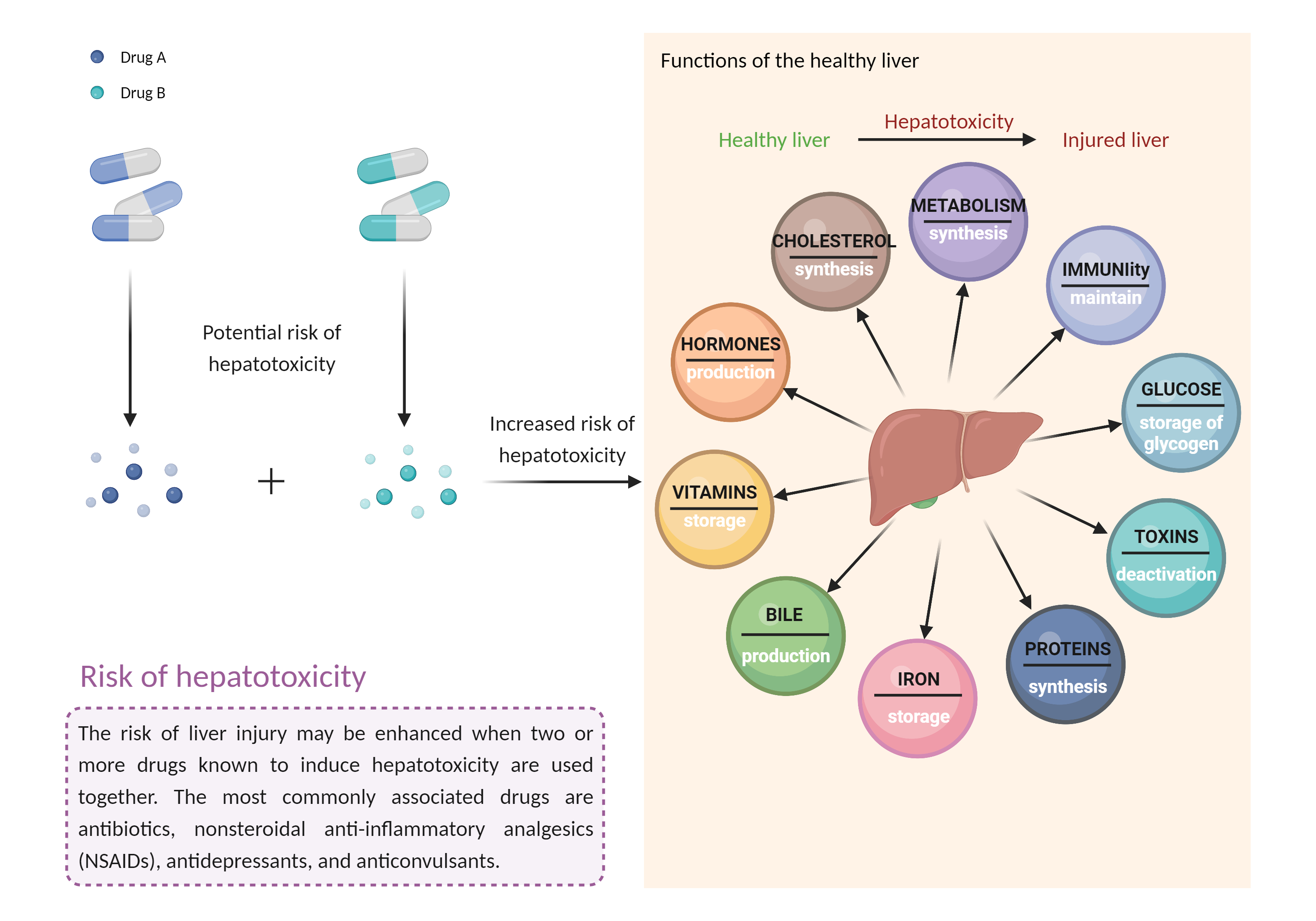
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cyclophosphamide | Azathioprine | |||||||
| Mechanism 2 | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | If concomitant therapy is needed patients should be monitored for signs and symptoms of hepatotoxicity and myelotoxicity. Patients should be advised to promptly notify their physician if they experience fever, chills, sore throat, mouth sores, unusual fatigue, bleeding, bruising, abdominal pain, darkened urine, or pale stools. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Shaunak S, Munro JM, Weinbren K, Walport MJ, Cox TM "Cyclophosphamide-induced liver necrosis: a possible interaction with azathioprine." Q J Med 67 (1988): 309-17. [PMID: 3060893] | ||||||||||||||||||
