Details of Drug-Drug Interaction
| Drug General Information (ID: DDIPUG68TQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ozanimod | Drug Info | Solriamfetol | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Selective Immunosuppressants | Cns Agents | |||||||
| Structure | |||||||||
| Mechanism of Ozanimod-Solriamfetol Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Additive dopaminergic effects Click to Show/Hide Mechanism Graph | |||||||||
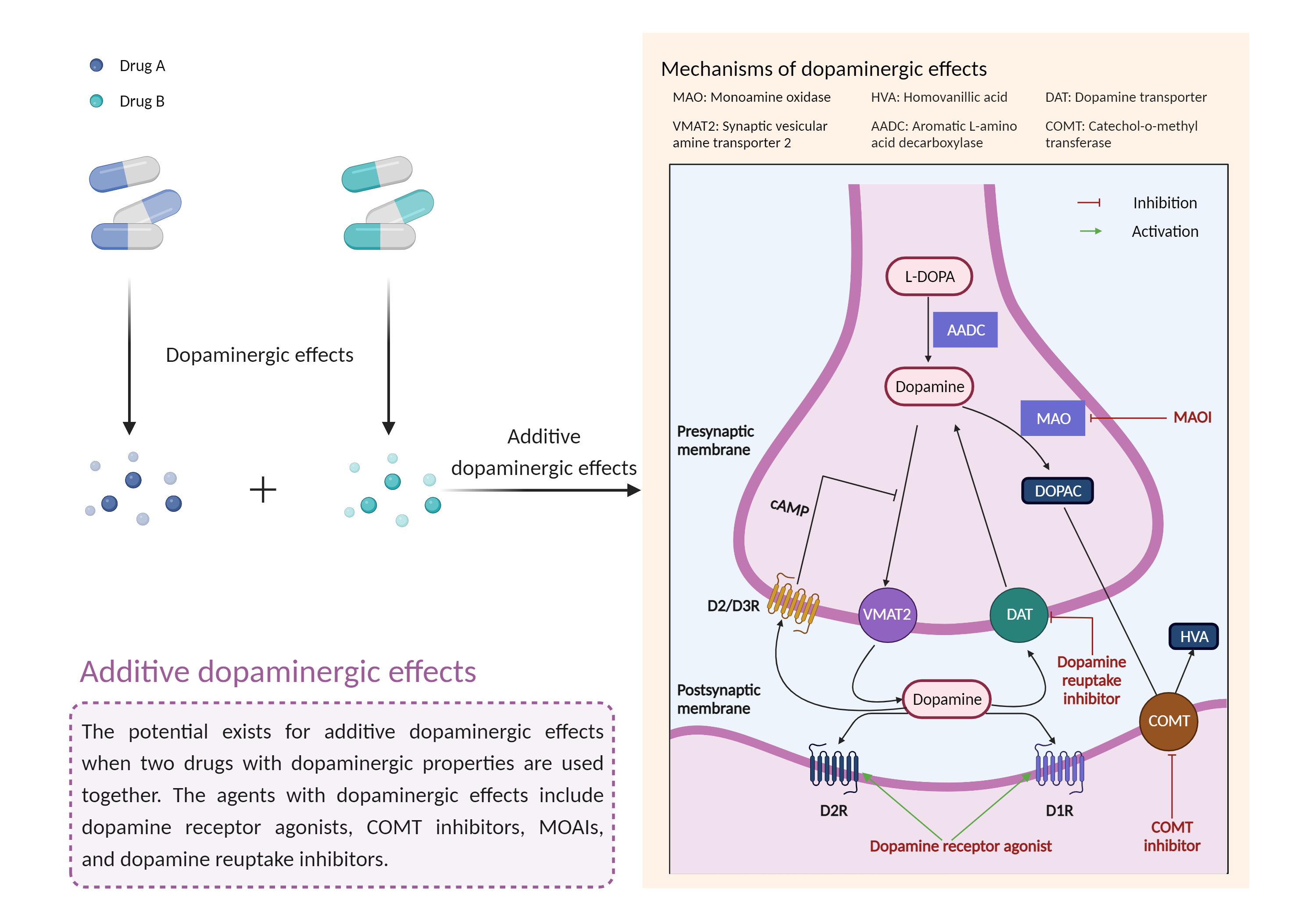 |
|||||||||
| Drug Name | Ozanimod | Solriamfetol | |||||||
| Mechanism 1 |
Dopaminergic effects Monoamine oxidase-B selective Inhibitor |
Dopaminergic effects Dopamine transporter Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Monoamine oxidase type B |
×
Structure
Sequence
MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSYVGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWRTMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEVSALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQTRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVYYKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEERLKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDRIYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTTFLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|||||||
| Gene Name | MAOB | ||||||||
| Uniprot ID | AOFB_HUMAN | ||||||||
| KEGG Pathway | hsa:4129 | ||||||||
| Protein Family | Flavin monoamine oxidase family | ||||||||
| Protein Function |
Catalyzes the oxidative deamination of primary and some secondary amines such as neurotransmitters, and exogenous amines including the tertiary amine, neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), with concomitant reduction of oxygen to hydrogen peroxide and participates in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079). Preferentially degrades benzylamine and phenylethylamine (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079).
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Dopamine transporter |
×
Structure
Sequence
MSKSKCSVGLMSSVVAPAKEPNAVGPKEVELILVKEQNGVQLTSSTLTNPRQSPVEAQDRETWGKKIDFLLSVIGFAVDLANVWRFPYLCYKNGGGAFLVPYLLFMVIAGMPLFYMELALGQFNREGAAGVWKICPILKGVGFTVILISLYVGFFYNVIIAWALHYLFSSFTTELPWIHCNNSWNSPNCSDAHPGDSSGDSSGLNDTFGTTPAAEYFERGVLHLHQSHGIDDLGPPRWQLTACLVLVIVLLYFSLWKGVKTSGKVVWITATMPYVVLTALLLRGVTLPGAIDGIRAYLSVDFYRLCEASVWIDAATQVCFSLGVGFGVLIAFSSYNKFTNNCYRDAIVTTSINSLTSFSSGFVVFSFLGYMAQKHSVPIGDVAKDGPGLIFIIYPEAIATLPLSSAWAVVFFIMLLTLGIDSAMGGMESVITGLIDEFQLLHRHRELFTLFIVLATFLLSLFCVTNGGIYVFTLLDHFAAGTSILFGVLIEAIGVAWFYGVGQFSDDIQQMTGQRPSLYWRLCWKLVSPCFLLFVVVVSIVTFRPPHYGAYIFPDWANALGWVIATSSMAMVPIYAAYKFCSLPGSFREKLAYAIAPEKDRELVDRGEVRQFTLRHWLKV
|
|||||||
| Gene Name | SLC6A3 | ||||||||
| Uniprot ID | SC6A3_HUMAN | ||||||||
| KEGG Pathway | hsa:6531 | ||||||||
| Protein Family | Sodium:neurotransmitter symporter (SNF) (TC 2.A.22) family | ||||||||
| Protein Function |
Amine transporter (PubMed:1406597, PubMed:8302271, PubMed:15505207). Terminates the action of dopamine by its high affinity sodium-dependent reuptake into presynaptic terminals (By similarity). Regulator of light-dependent retinal hyaloid vessel regression, downstream of OPN5 signaling (By similarity).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Additive hypertensive effects Click to Show/Hide Mechanism Graph | |||||||||
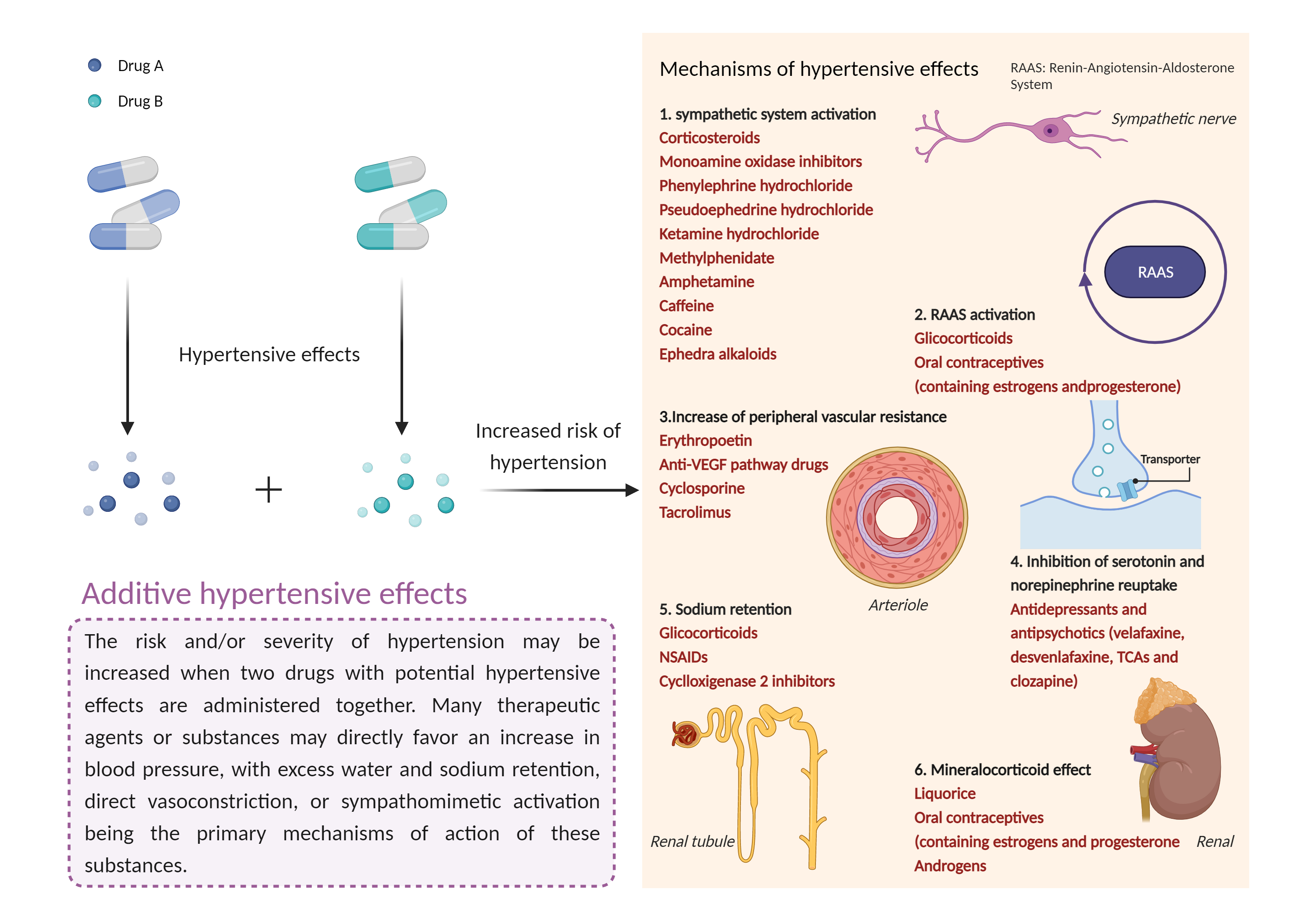 |
|||||||||
| Drug Name | Ozanimod | Solriamfetol | |||||||
| Mechanism 2 | Hypertensive effects | Hypertensive effects | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Hypertensive effects | ||||||||
| Factor Description | Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of solriamfetol with MAOIs is considered contraindicated. At least 14 days should elapse between discontinuation of MAOI therapy and initiation of treatment with solriamfetol. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Sunosi (solriamfetol). Jazz Pharmaceuticals, Palo Alto, CA. | ||||||||||||||||||
