| Management |
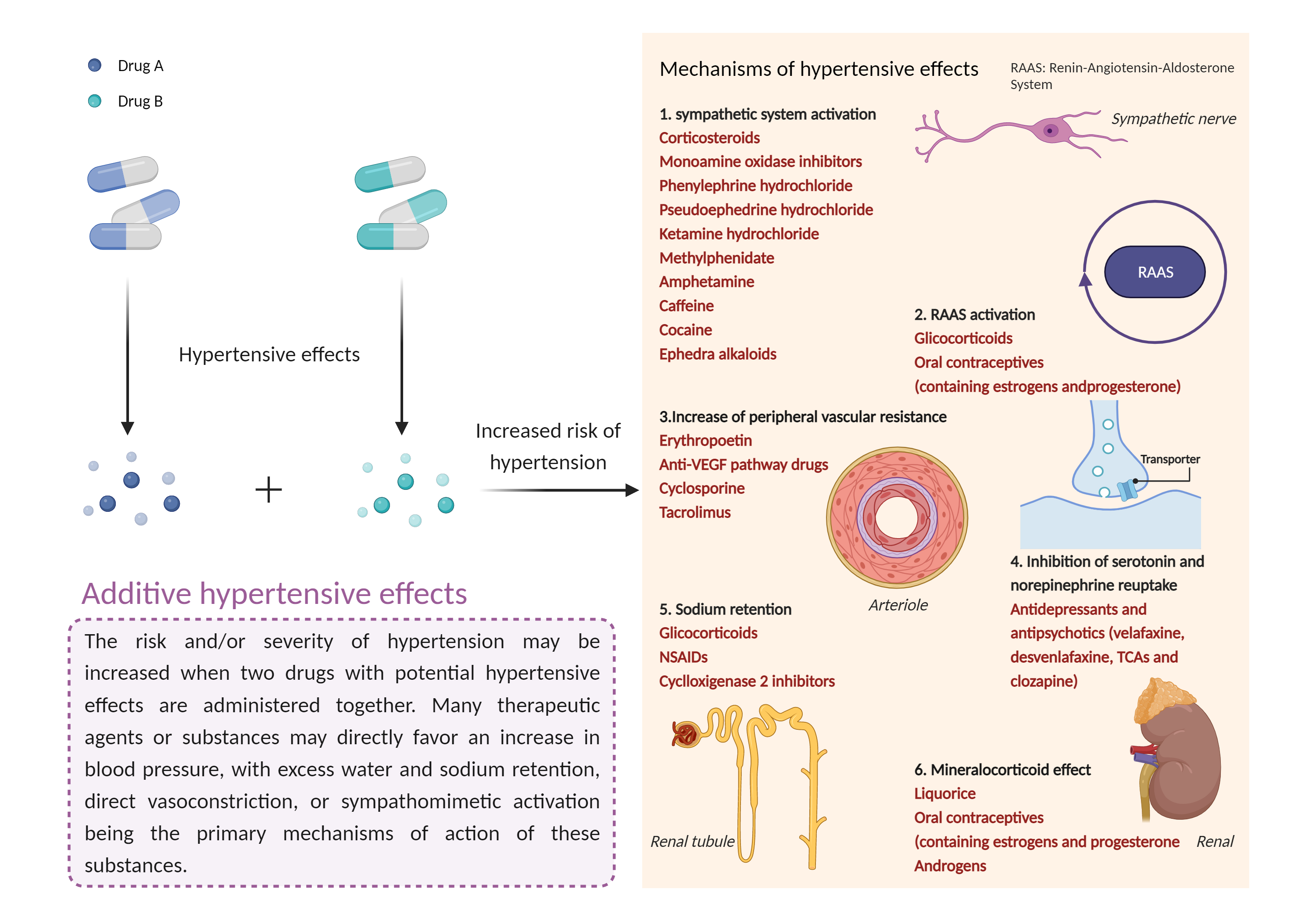
Caution is advised and blood pressure should be closely monitored during concomitant use of esketamine with psychostimulants or MAOIs. All patients receiving esketamine should have BP assessed prior to administration. If BP is elevated (e.g., >140 mmHg systolic, >90 mmHg diastolic), a delay in esketamine treatment may be necessary, taking into consideration the benefits versus risks in individual patients. BP should be monitored for at least 2 hours after esketamine administration, starting at approximately 40 minutes post-dose and subsequently as clinically warranted. In patients with a history of hypertensive encephalopathy, more intensive monitoring is warranted due to increased risk for developing encephalopathy with even small increases in BP. If at any point BP is elevated and remains high, promptly seek assistance from practitioners experienced in BP management. Patients experiencing symptoms of a hypertensive crisis (e.g., chest pain, shortness of breath) or hypertensive encephalopathy (e.g., sudden severe headache, visual disturbances, seizures, diminished consciousness or focal neurological deficits) should be immediately referred for emergency care. |