Details of Drug-Drug Interaction
| Drug General Information (ID: DDIODY82CX) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Etidronic acid | Drug Info | Remdesivir | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Bone Density Conservation Agents | Purine Nucleosides | |||||||
| Structure | |||||||||
| Mechanism of Etidronic acid-Remdesivir Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
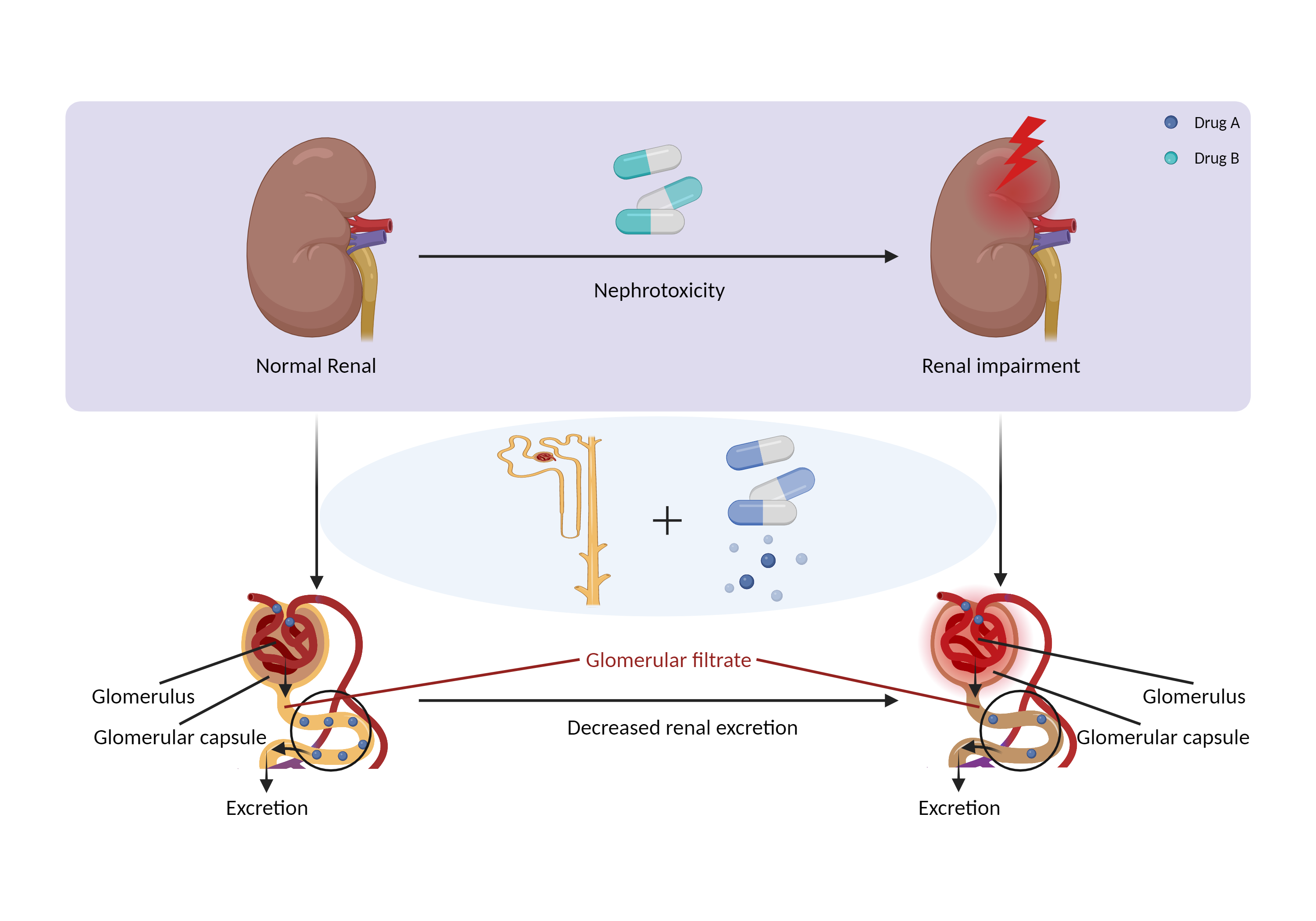
| Decreased renal excretion due to nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Etidronic acid | Remdesivir | |||||||
| Mechanism | Nephrotoxicity | Primarily eliminated by renal excretion | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | Drug-induced nephrotoxicity is a common and potentially serious complication of medication use. The kidneys are the primary organ for drug excretion, and when they are damaged can lead to decreased drug excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Some authorities recommend avoiding the concomitant use of remdesivir with drugs that can reduce renal function. Caution is advised if remdesivir is used in patients who have recently received potentially nephrotoxic agents (e.g., aminoglycosides polypeptide, glycopeptide, and polymyxin antibiotics amphotericin B adefovir cidofovir tenofovir foscarnet cisplatin deferasirox gallium nitrate lithium mesalamine certain immunosuppressants intravenous bisphosphonates intravenous pentamidine high intravenous dosages of methotrexate high dosages and/or chronic use of nonsteroidal anti-inflammatory agents). Renal function (estimated glomerular filtration rate, or eGFR, for adult and pediatric patients older than 28 days serum creatinine for full-term neonates at least 7 days up to 28 days old) should be evaluated prior to starting remdesivir and monitored daily during treatment or as clinically appropriate. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | European Medicines Agency "Summary on compassionate use. Remdesivir Gilead.". | ||||||||||||||||||
| 3 | Gilead Sciences, Inc "About Remdesivir.". | ||||||||||||||||||
