Details of Drug-Drug Interaction
| Drug General Information (ID: DDIO8H521D) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Methotrexate | Drug Info | Probenecid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antimetabolites | Uricosuric Agents | |||||||
| Structure | |||||||||
| Mechanism of Methotrexate-Probenecid Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
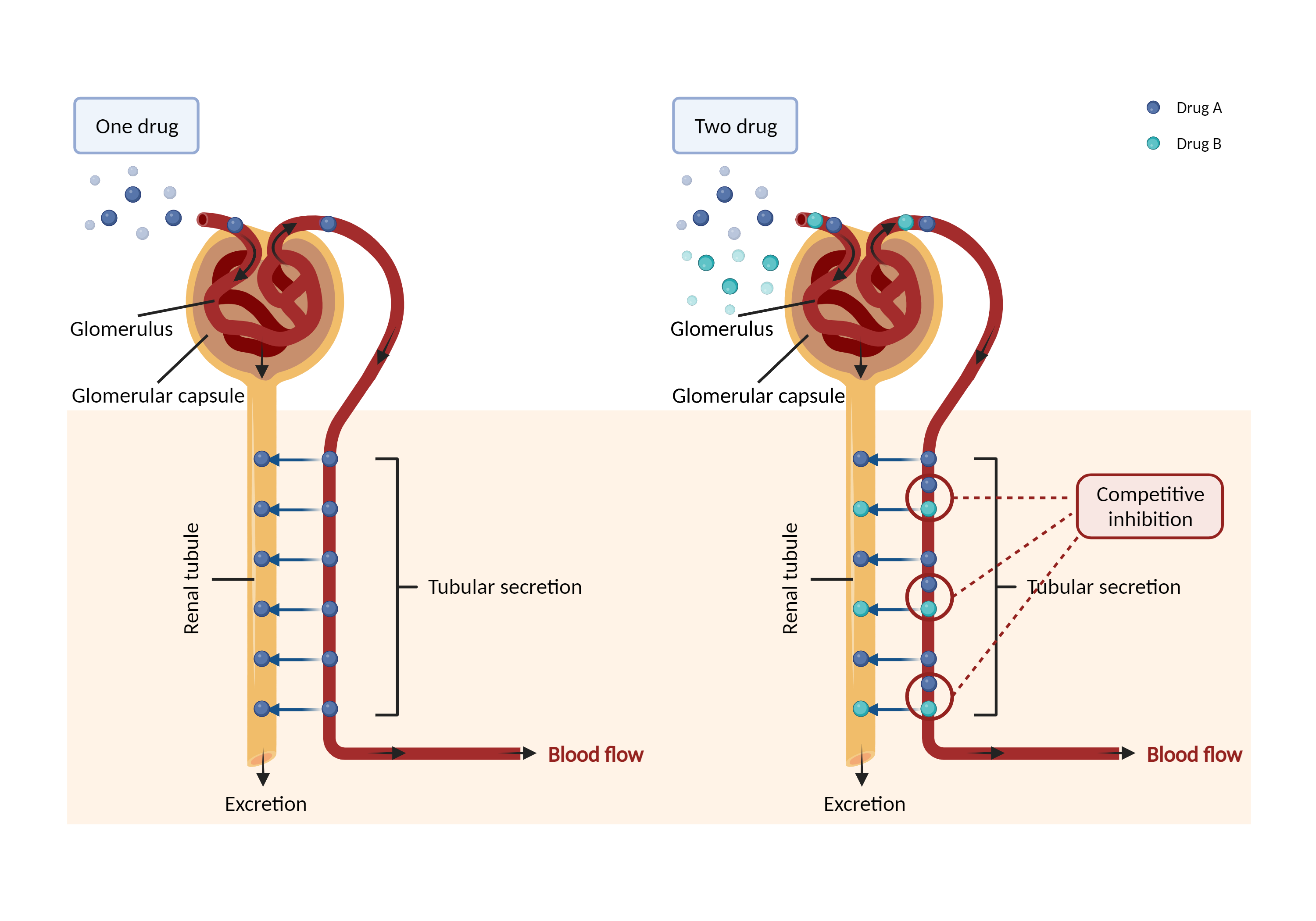
| Competitive inhibition of renal tubular secretion Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Methotrexate | Probenecid | |||||||
| Mechanism | Methotrexate | Competitive inhibition of renal tubular secretion of methotrexate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Renal tubular secretion | ||||||||
| Factor Description | Renal tubular secretion allows selective transfer of substances from the blood around the capillaries into the renal tubules via filtrate. Drug excretion may be reduced when two drugs compete for renal tubular secretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | If this combination must be used, a reduction in methotrexate dosage may be needed and the patient should be closely monitored for signs and symptoms of bone marrow suppression, hepatotoxicity, and nephrotoxicity. Patients should be advised to promptly report symptoms including fever, chills, sore throat, bruising, bleeding, stomatitis, malaise, shortness of breath, lower extremity edema, jaundice, or change in stool or urine color to their physician. | ||||||||
