Details of Drug-Drug Interaction
| Drug General Information (ID: DDINS9Z1HX) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Entecavir | Drug Info | Mefenamic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiviral Agents | Antiinflammatory Agents | |||||||
| Structure | |||||||||
| Mechanism of Entecavir-Mefenamic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
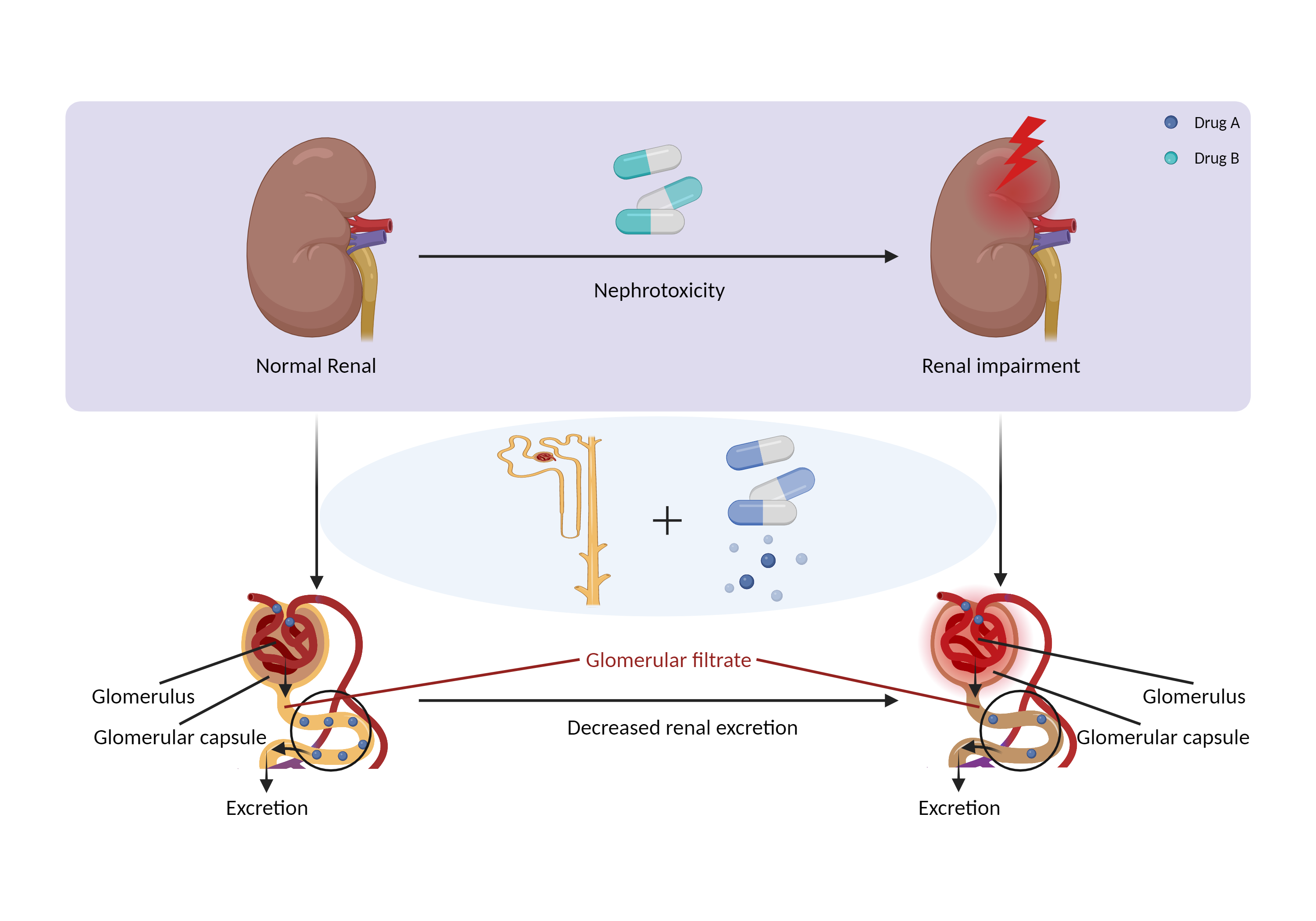
| Decreased renal excretion due to nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Entecavir | Mefenamic acid | |||||||
| Mechanism | Primarily eliminated by renal excretion | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | Drug-induced nephrotoxicity is a common and potentially serious complication of medication use. The kidneys are the primary organ for drug excretion, and when they are damaged can lead to decreased drug excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if entecavir is prescribed in combination with potentially nephrotoxic drugs (e.g., aminoglycosides polypeptide, glycopeptide, and polymyxin antibiotics amphotericin B adefovir cidofovir tenofovir foscarnet cisplatin deferasirox gallium nitrate lithium mesalamine certain immunosuppressants intravenous bisphosphonates intravenous pentamidine high intravenous dosages of methotrexate high dosages and/or chronic use of nonsteroidal anti-inflammatory agents). Renal function should be evaluated prior to and during therapy with entecavir. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Baraclude (entecavir). Bristol-Myers Squibb, Princeton, NJ. | ||||||||||||||||||
