| Management |
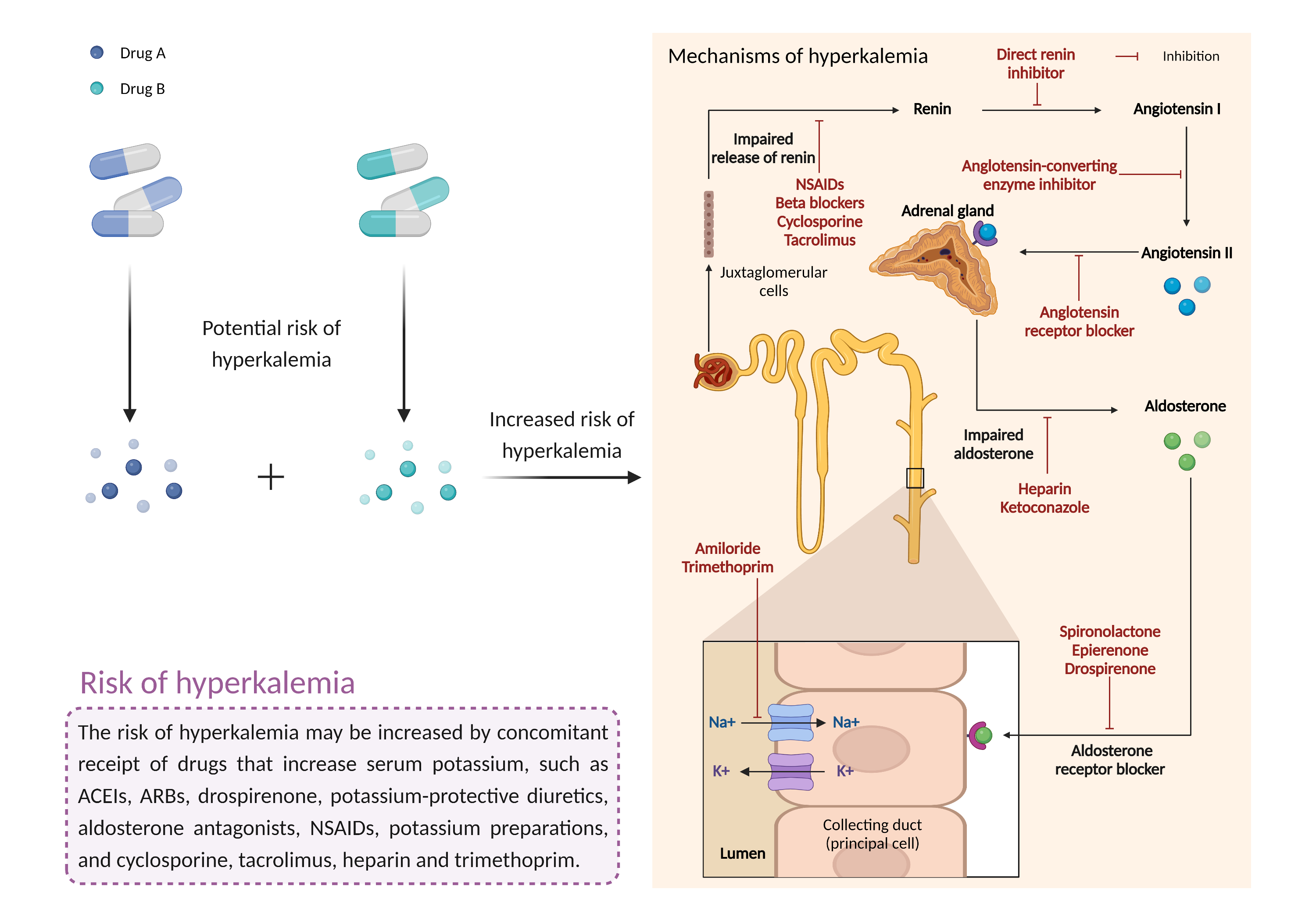
The use of aliskiren with ACE inhibitors or ARBs is considered contraindicated in patients with diabetes and should be avoided in general, particularly in patients with moderate to severe renal impairment (i.e., creatinine clearance (CrCl) < 60 mL/min). Prescribers should not initiate aliskiren in diabetic patients who are taking an ACE inhibitor or an ARB, and should stop any aliskiren-containing treatment if these patients are already receiving the combination. alternative antihypertensive treatment should be considered as necessary. Most patients do not obtain any additional benefit from combination therapy relative to monotherapy therefore, the potential risks should be thoroughly assessed when aliskiren is prescribed with ACE inhibitors or ARBs for the treatment of essential hypertension in patients without diabetes. Volume or salt depletion should be corrected prior to initiation of treatment. Routine monitoring of blood pressure, electrolytes, and renal function are recommended, particularly in the elderly or patients with worsening heart failure or a risk for dehydration. Potassium supplementation should generally be avoided unless it is closely monitored, and patients should be advised to seek medical attention if they experience signs and symptoms of hyperkalemia such as weakness, listlessness, confusion, tingling of the extremities, and irregular heartbeat. |