Details of Drug-Drug Interaction
| Drug General Information (ID: DDINGU4ELC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Tetrabenazine | Drug Info | Deutetrabenazine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Nephropathic Cystinosis Therapy | Vmat2 Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Tetrabenazine-Deutetrabenazine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
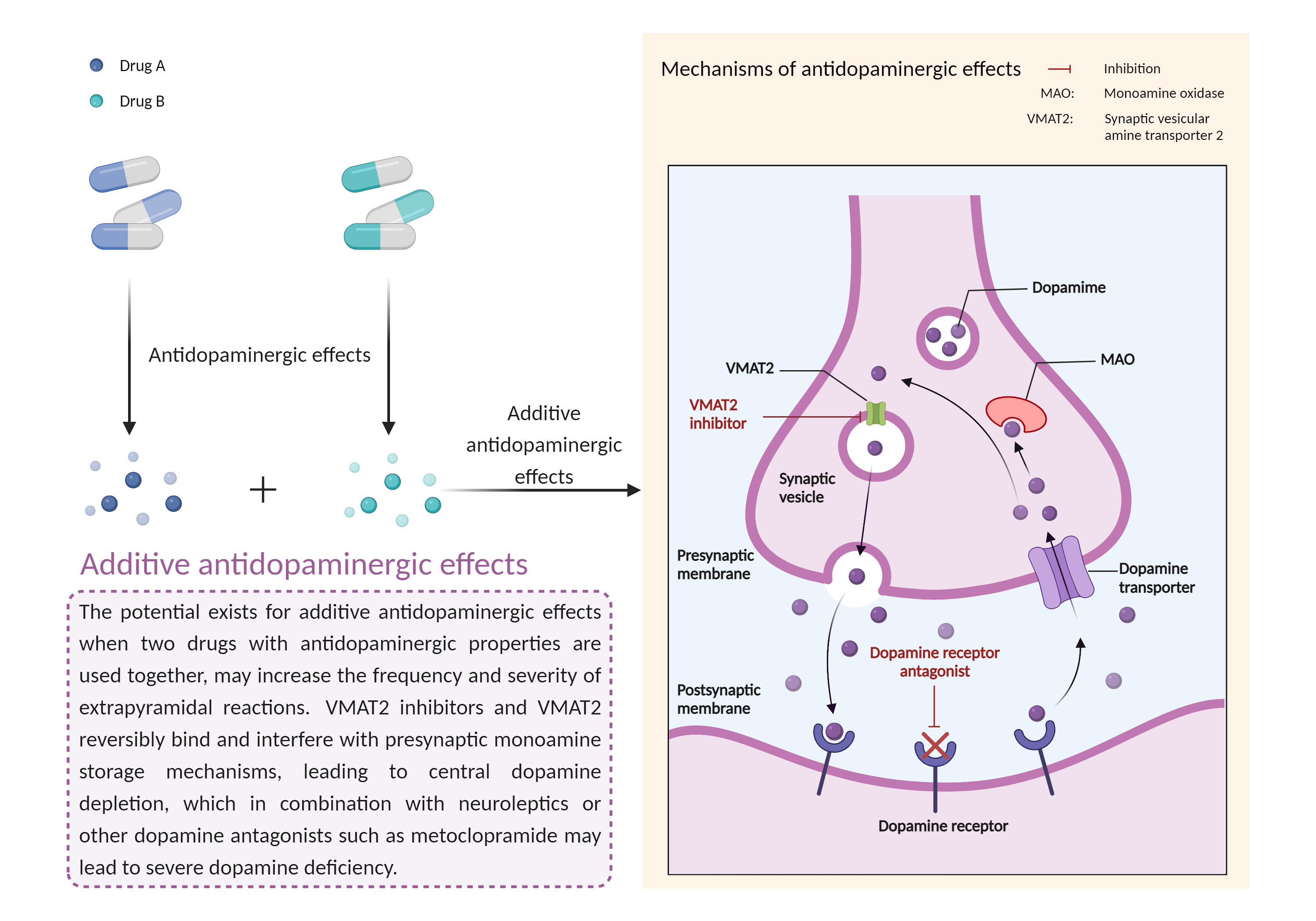
| Additive antidopaminergic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Tetrabenazine | Deutetrabenazine | |||||||
| Mechanism |
Antidopaminergic effects Synaptic vesicular amine transporter Inhibitor |
Antidopaminergic effects Synaptic vesicular amine transporter Inhibitor |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Synaptic vesicle amine transporter |
×
Structure
Sequence
MALSELALVRWLQESRRSRKLILFIVFLALLLDNMLLTVVVPIIPSYLYSIKHEKNATEIQTARPVHTASISDSFQSIFSYYDNSTMVTGNATRDLTLHQTATQHMVTNASAVPSDCPSEDKDLLNENVQVGLLFASKATVQLITNPFIGLLTNRIGYPIPIFAGFCIMFVSTIMFAFSSSYAFLLIARSLQGIGSSCSSVAGMGMLASVYTDDEERGNVMGIALGGLAMGVLVGPPFGSVLYEFVGKTAPFLVLAALVLLDGAIQLFVLQPSRVQPESQKGTPLTTLLKDPYILIAAGSICFANMGIAMLEPALPIWMMETMCSRKWQLGVAFLPASISYLIGTNIFGILAHKMGRWLCALLGMIIVGVSILCIPFAKNIYGLIAPNFGVGFAIGMVDSSMMPIMGYLVDLRHVSVYGSVYAIADVAFCMGYAIGPSAGGAIAKAIGFPWLMTIIGIIDILFAPLCFFLRSPPAKEEKMAILMDHNCPIKTKMYTQNNIQSYPIGEDEESESD
|
|||||||
| Gene Name | SLC18A2 | ||||||||
| Uniprot ID | VMAT2_HUMAN | ||||||||
| KEGG Pathway | hsa:6571 | ||||||||
| Protein Family | Major facilitator superfamily | ||||||||
| Protein Function |
Involved in the ATP-dependent vesicular transport of biogenic amine neurotransmitters. Pumps cytosolic monoamines including dopamine, norepinephrine, serotonin, and histamine into synaptic vesicles (PubMed:23363473). Requisite for vesicular amine storage prior to secretion via exocytosis.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of tetrabenazine and deutetrabenazine is considered contraindicated. Treatment with deutetrabenazine may be started the day following discontinuation of tetrabenazine. The manufacturer's product labeling should be consulted for dosing guidelines when switching from tetrabenazine to deutetrabenazine. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Austedo (deutetrabenazine). Teva Pharmaceuticals USA, North Wales, PA. | ||||||||||||||||||
