| Management |
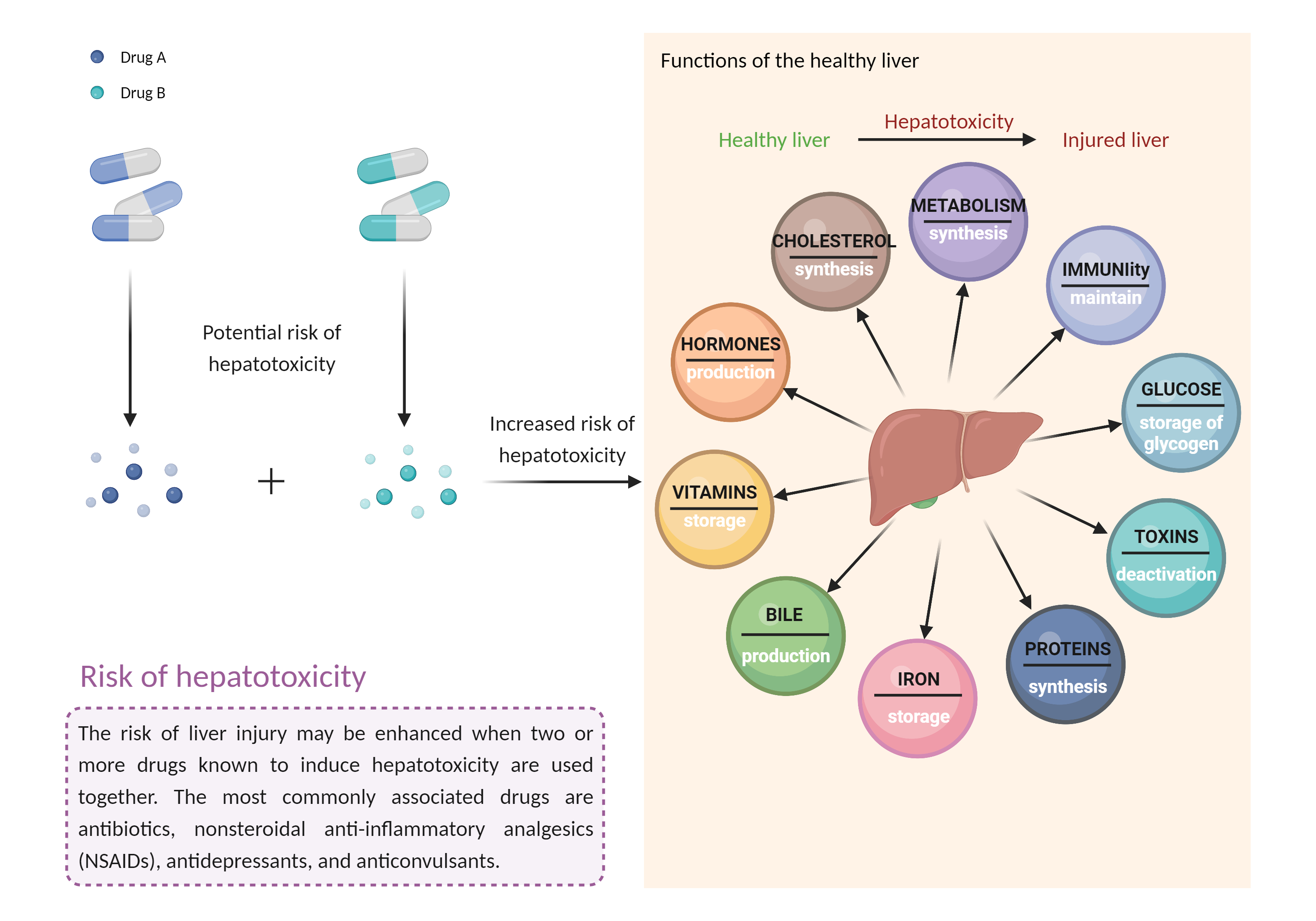
The use of idelalisib with other potentially hepatotoxic agents such as azole antifungal agents, macrolide antibiotics, nefazodone, ritonavir, and telithromycin should be avoided whenever possible. In addition, these agents are potent CYP450 3A4/P-gp inhibitors and may increase the toxicity of idelalisib. Caution is advised if coadministration is required. Patients should be closely monitored for hepatotoxicity and other toxicities of idelalisib such as diarrhea, colitis, intestinal perforation, pneumonitis, neutropenia and thrombocytopenia, and the dosing adjusted or interrupted as necessary. Patients should have serum ALT, AST, and bilirubin measured prior to initiation of treatment and regularly during treatment in accordance with the product labeling. Permanent discontinuation of idelalisib is recommended in those who experience recurrent hepatotoxicity following dosage reduction. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hepatotoxicity such as fever, rash, itching, anorexia, nausea, vomiting, fatigue, malaise, right upper quadrant pain, dark urine, pale stools, and jaundice. |