Details of Drug-Drug Interaction
| Drug General Information (ID: DDIMXLP3CY) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Bacampicillin | Drug Info | Magnesium carbonate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antibiotics | Antacids | |||||||
| Structure | |||||||||
| Mechanism of Bacampicillin-Magnesium carbonate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
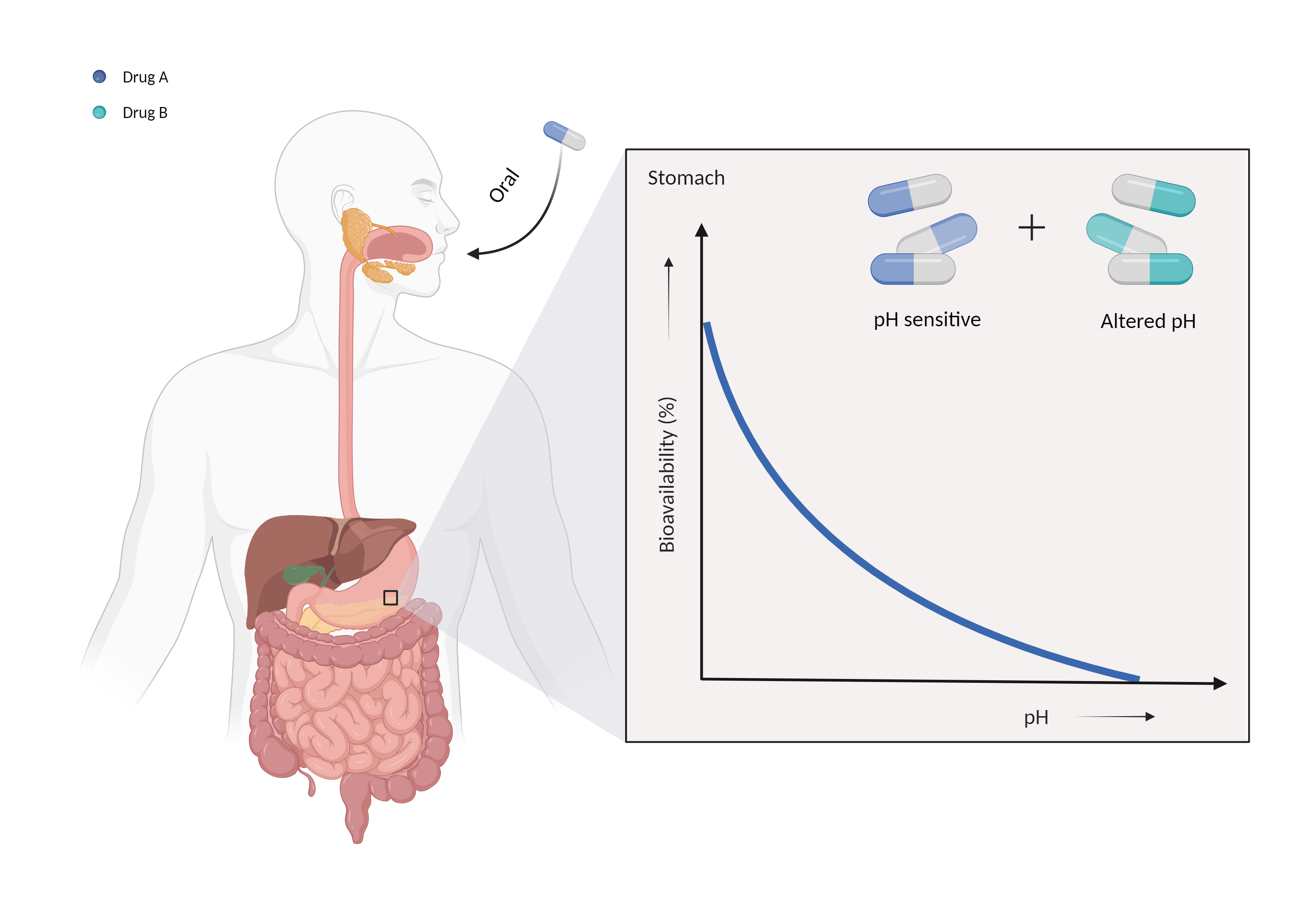
| Altered gastric pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Bacampicillin | Magnesium carbonate | |||||||
| Mechanism | Gastric pH sensitive | Gastric alkalinizer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastric pH | ||||||||
| Factor Description | The normal pH range of gastric acid is between 1.5 and 3.5 and is highly acidic, consisting mainly of hydrochloric acid. Changes in the pH of the stomach can alter the absorption of drugs. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The possibility of a reduced or subtherapeutic response to bacampicillin should be considered during coadministration with antacids, H2-receptor antagonists, proton pump inhibitors, or other agents that can increase gastric pH. Preferably, these agents should be avoided during therapy with bacampicillin, or an alternative antibiotic be prescribed if these medications cannot be discontinued. Patients treated with antacids (or oral medications that contain antacids such as didanosine buffered tablets or pediatric oral solution) may minimize the effects of the interaction with bacampicillin by separating the times of administration by at least 2 hours. | ||||||||
