Details of Drug-Drug Interaction
| Drug General Information (ID: DDIMXF48HG) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Linezolid | Drug Info | Safinamide | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antibiotics | Dopaminergic Antiparkinsonism Agents | |||||||
| Structure | |||||||||
| Mechanism of Linezolid-Safinamide Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
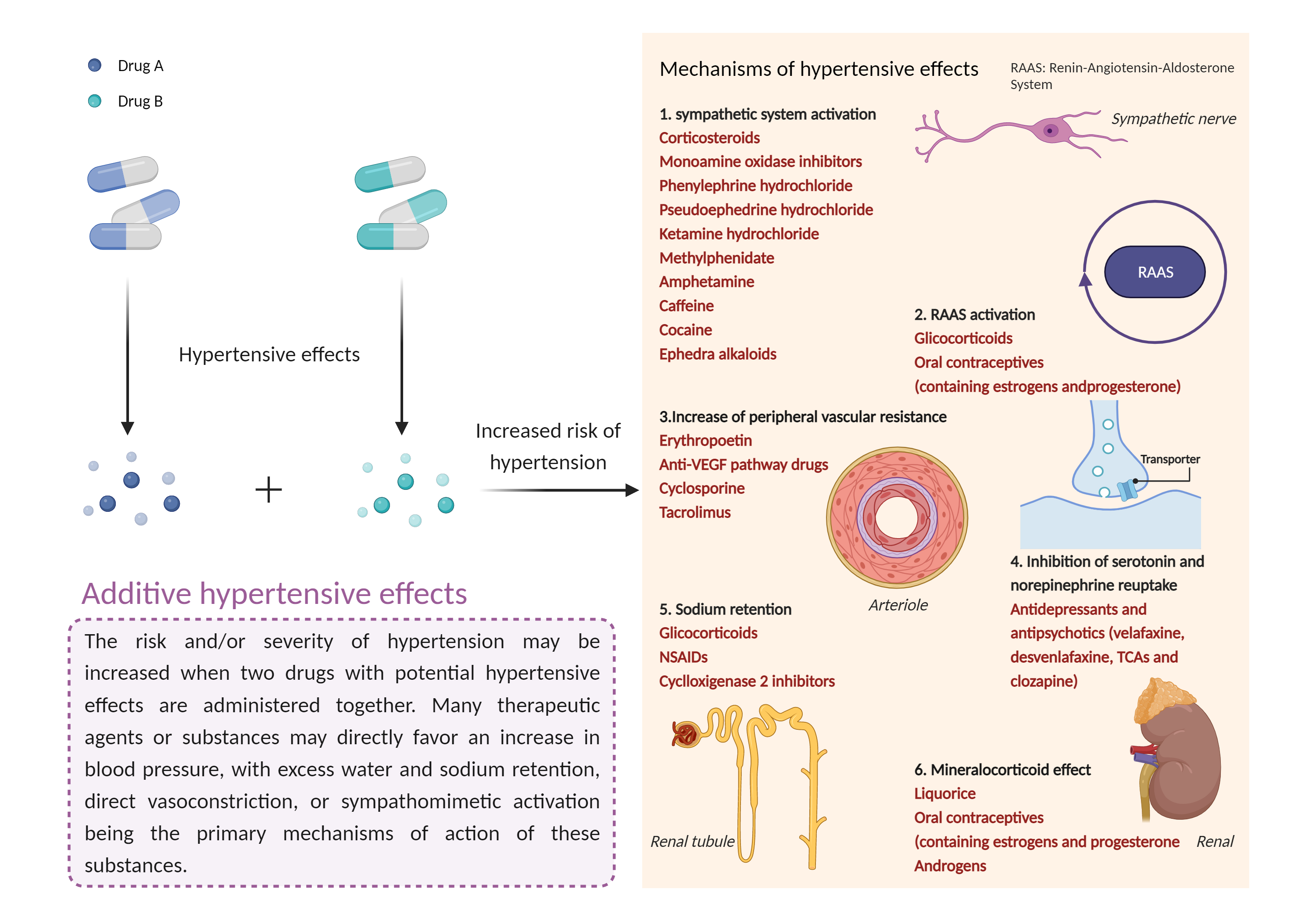
| Additive hypertensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Linezolid | Safinamide | |||||||
| Mechanism 1 | Hypertensive effects | Hypertensive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypertensive effects | ||||||||
| Factor Description | Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. | ||||||||
| Mechanism Description |
|
||||||||
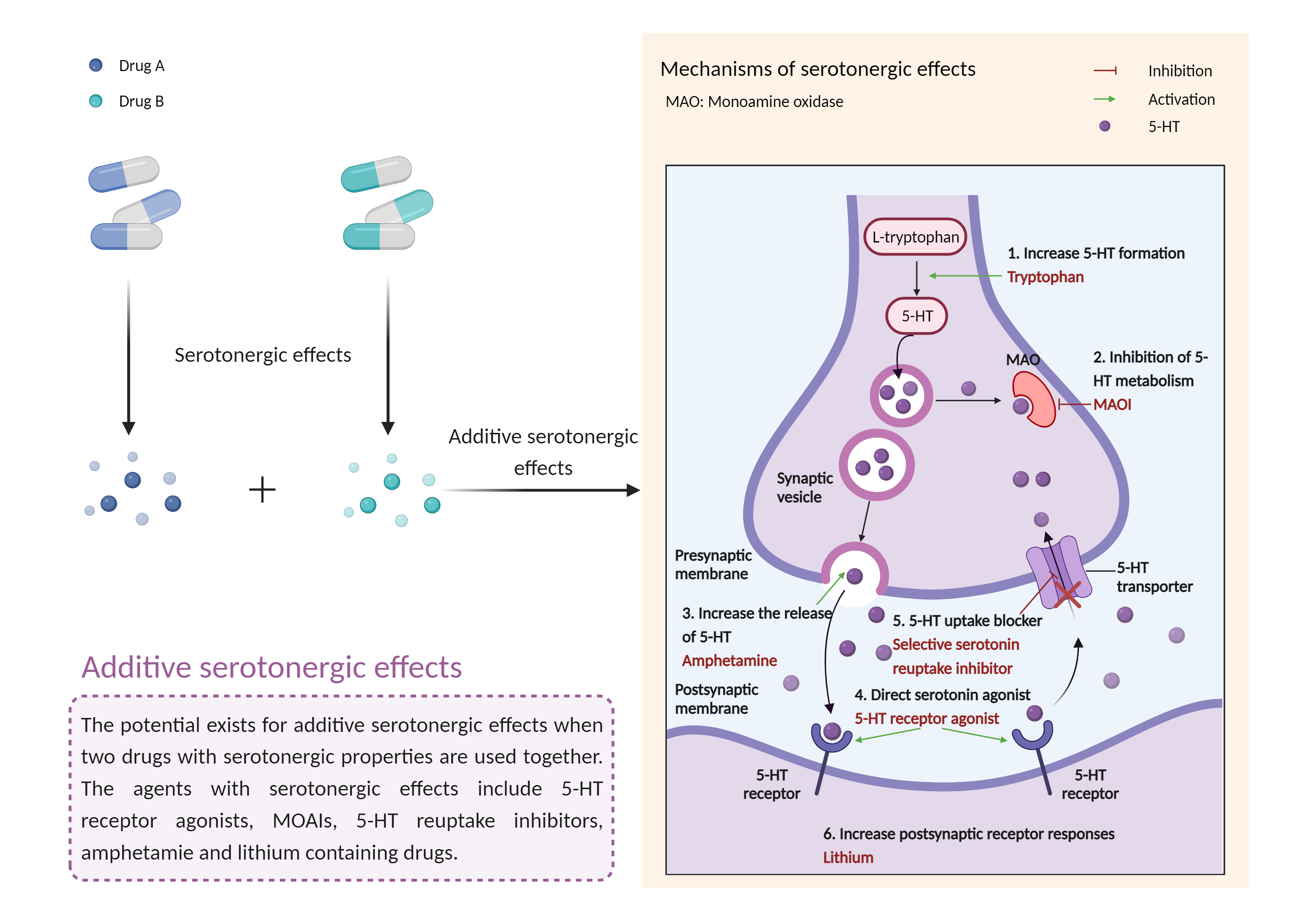
| Additive serotonergic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Linezolid | Safinamide | |||||||
| Mechanism 2 |
Serotonergic effects Monoamine oxidase non-selective Inhibitor |
Serotonergic effects Monoamine oxidase-B selective Inhibitor |
|||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Monoamine oxidase | Structure Sequence | |||||||
| Protein Family | Flavin monoamine oxidase family | ||||||||
| Protein Function |
Catalyzes the oxidative deamination of primary and some secondary amine such as neurotransmitters, with concomitant reduction of oxygen to hydrogen peroxide and has important functions in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues (PubMed:20493079, PubMed:8316221, PubMed:18391214, PubMed:24169519). Preferentially oxidizes serotonin (PubMed:20493079, PubMed:24169519). Also catalyzes the oxidative deamination of kynuramine to 3-(2-aminophenyl)-3-oxopropanal that can spontaneously condense to 4-hydroxyquinoline (By similarity).
Click to Show/Hide
|
||||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Monoamine oxidase type B |
×
Structure
Sequence
MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSYVGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWRTMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEVSALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQTRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVYYKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEERLKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDRIYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTTFLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|||||||
| Gene Name | MAOB | ||||||||
| Uniprot ID | AOFB_HUMAN | ||||||||
| KEGG Pathway | hsa:4129 | ||||||||
| Protein Family | Flavin monoamine oxidase family | ||||||||
| Protein Function |
Catalyzes the oxidative deamination of primary and some secondary amines such as neurotransmitters, and exogenous amines including the tertiary amine, neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), with concomitant reduction of oxygen to hydrogen peroxide and participates in the metabolism of neuroactive and vasoactive amines in the central nervous system and peripheral tissues (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079). Preferentially degrades benzylamine and phenylethylamine (PubMed:11134050, PubMed:8665924, PubMed:8316221, PubMed:11049757, PubMed:20493079).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of linezolid concurrently or within two weeks of taking any drug that inhibits monoamine oxidase A or B is generally considered contraindicated. In emergency situations requiring urgent treatment with linezolid (e.g., vancomycin-resistant Enterococcus faecium (VRE) infections nosocomial pneumonia and complicated skin and skin structure infections, including cases caused by methicillin-resistant Staphylococcus aureus), the availability of alternative interventions should be considered in patients already receiving a MAOI and the benefit of linezolid treatment should be weighed against the risk of serotonin toxicity. If linezolid is required, the MAOI must be immediately stopped, and the patient closely monitored for emergent symptoms of CNS toxicity for two weeks (five weeks if fluoxetine was taken) or until 24 hours after the last dose of linezolid, whichever comes first. Patients and/or their caregivers should be advised to seek medical attention if potential symptoms of serotonin syndrome develop. Treatment with the MAOI may be resumed 24 hours after the last dose of linezolid. | ||||||||
