Details of Drug-Drug Interaction
| Drug General Information (ID: DDIMVHU7D6) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Bacillus calmette-guerin substrain tice live antigen | Drug Info | Human botulinum neurotoxin A/B immune globulin | Drug Info | |||||
| Drug Type | Vaccine | Polyclonal antibody | |||||||
| Therapeutic Class | Vaccine | Immune Globulins | |||||||
| Mechanism of Bacillus calmette-guerin substrain tice live antigen-Human botulinum neurotoxin A/B immune globulin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
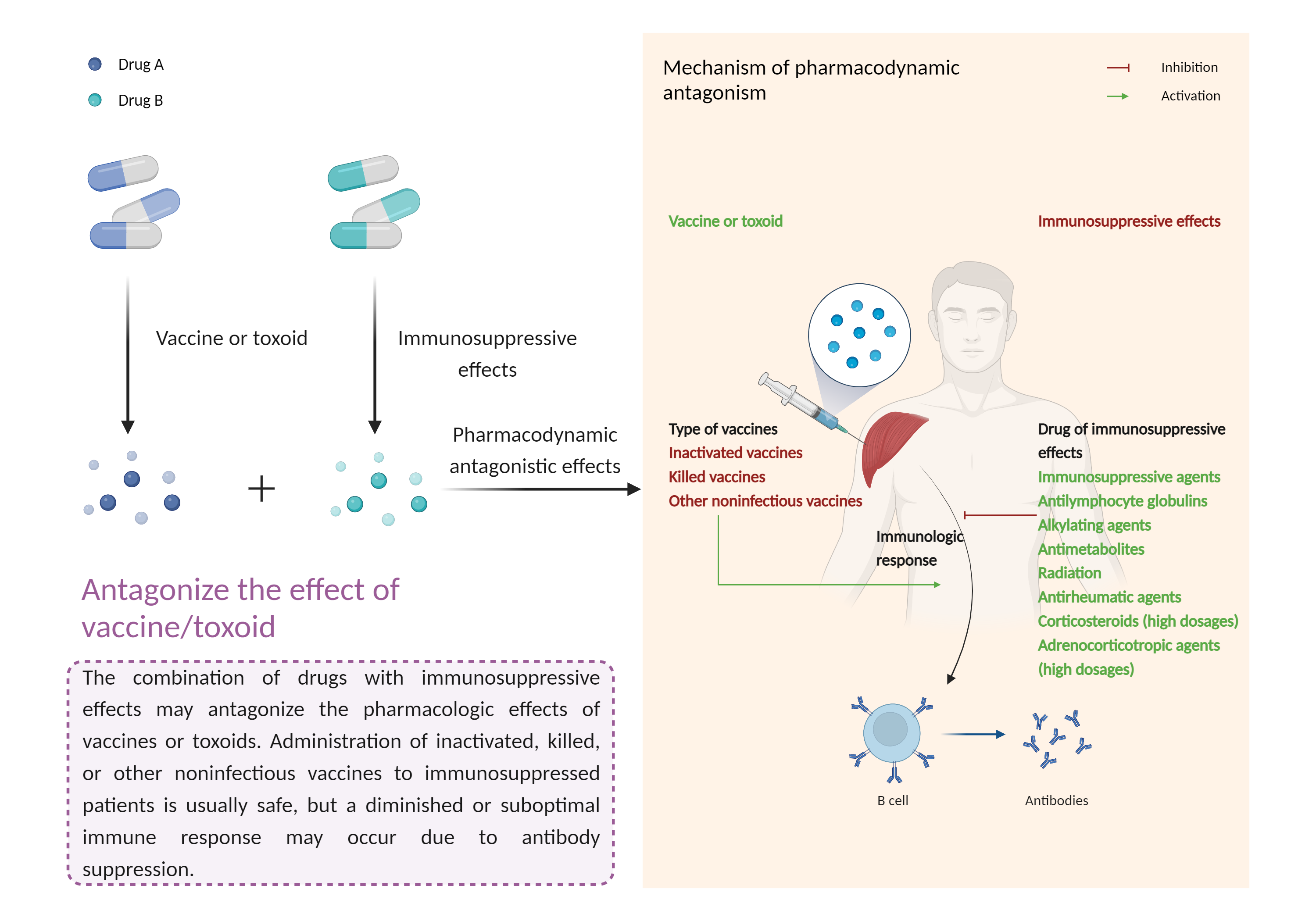
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Bacillus calmette-guerin substrain tice live antigen | Human botulinum neurotoxin A/B immune globulin | |||||||
| Mechanism | Vaccine or toxoid | Immunomodulatory effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Varicella virus vaccine should not be given for at least 5 months, and other live vaccines should not be given for at least 3 months, after administration of immune globulin. If such vaccinations are given shortly before or after immune globulin therapy, a revaccination may be necessary. | ||||||||
