Details of Drug-Drug Interaction
| Drug General Information (ID: DDIMF28DHJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Potassium citrate | Drug Info | Homatropine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Minerals And Electrolytes | Mydriatics | |||||||
| Structure | |||||||||
| Mechanism of Potassium citrate-Homatropine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
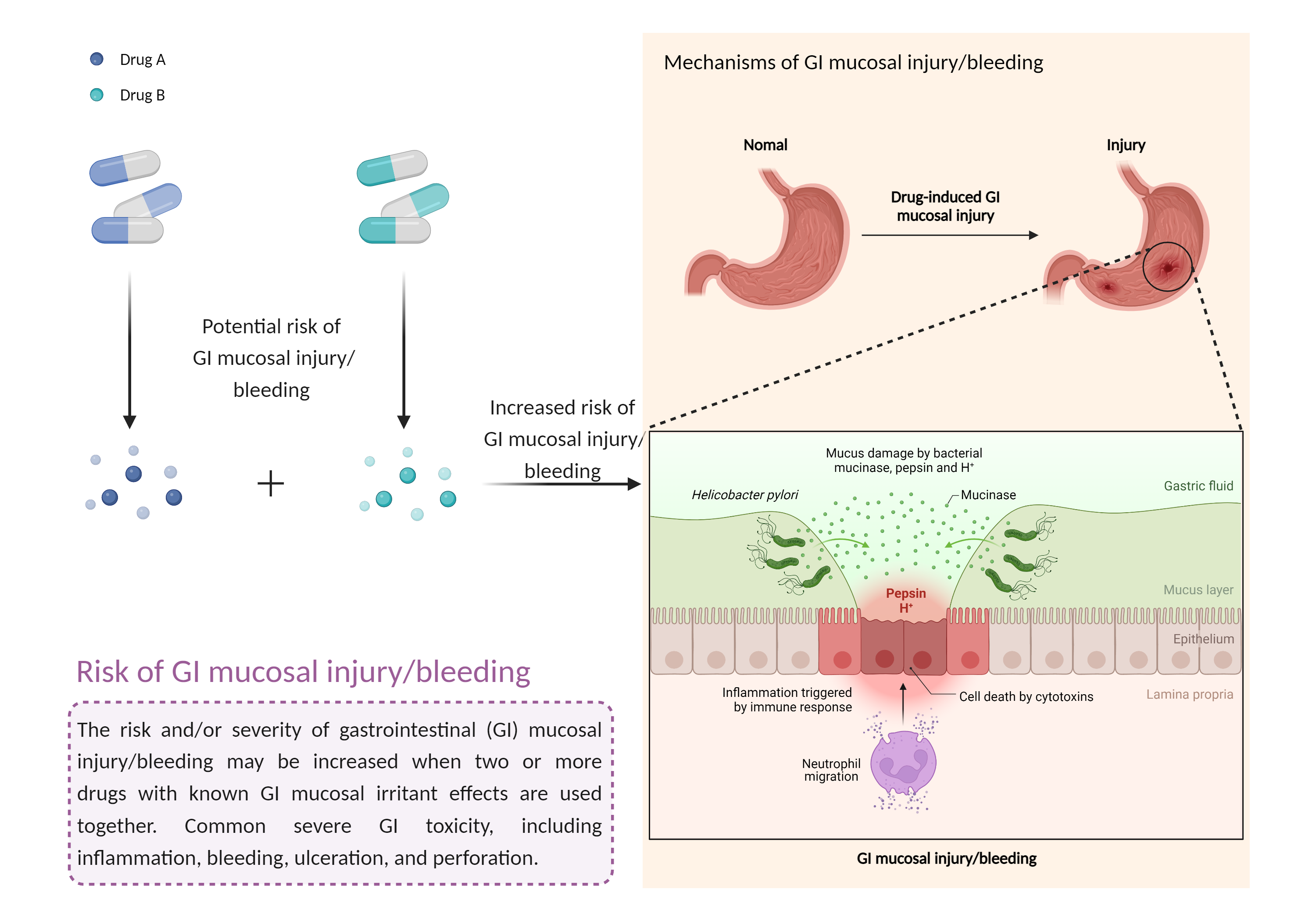
| Increased risk of GI mucosal injury/bleeding risk Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Potassium citrate | Homatropine | |||||||
| Mechanism | GI mucosal injury/bleeding risk | Decrease gastrointestinal motility | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal toxicity | ||||||||
| Factor Description | The risk and severity of gastrointestinal toxicity, including inflammation, bleeding, ulceration, and perforation, increases when drugs with gastrointestinal mucosal irritant effects are combined. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of oral solid formulations of potassium citrate is considered contraindicated in patients receiving agents with anticholinergic properties at sufficient doses to exert anticholinergic effects. A liquid formulation of potassium citrate should be considered. Patients prescribed a solid oral formulation should be advised to discontinue potassium therapy and contact their physician if they experience potential symptoms of upper GI injury such as severe vomiting, abdominal pain, distention, and gastrointestinal bleeding. | ||||||||
