Details of Drug-Drug Interaction
| Drug General Information (ID: DDIMD5WLXC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Safinamide | Drug Info | Vilanterol | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Dopaminergic Antiparkinsonism Agents | Bronchodilators | |||||||
| Structure | |||||||||
| Mechanism of Safinamide-Vilanterol Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
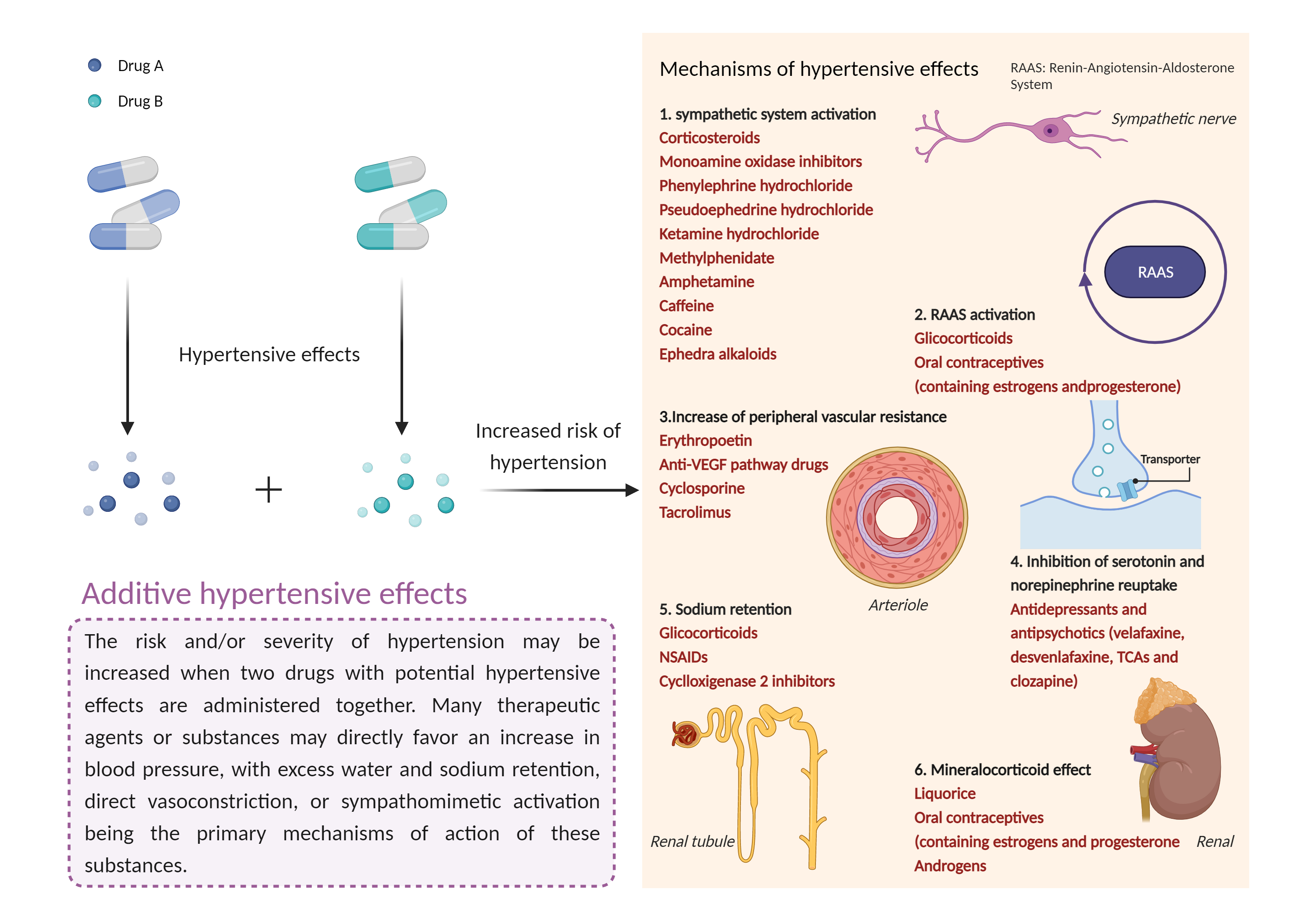
| Additive hypertensive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Safinamide | Vilanterol | |||||||
| Mechanism | Hypertensive effects | Hypertensive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypertensive effects | ||||||||
| Factor Description | Hypertension is defined as a blood pressure reading above a prescribed limit (130/90 mmHg). Most people with hypertension have no signs or symptoms; others may experience: severe headache, shortness of breath, nosebleeds, severe anxiety, and feeling a tingling sensation in the neck or head. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Cardiovascular status should be closely monitored when beta-2 agonists are coadministered with MAOIs or other agents that possess MAOI activity (e.g., furazolidone, linezolid, methylene blue, procarbazine). Preferably, at least 14 days should elapse between discontinuation of MAOI therapy and initiation of treatment with beta-2 agonists. | ||||||||
