| Management |
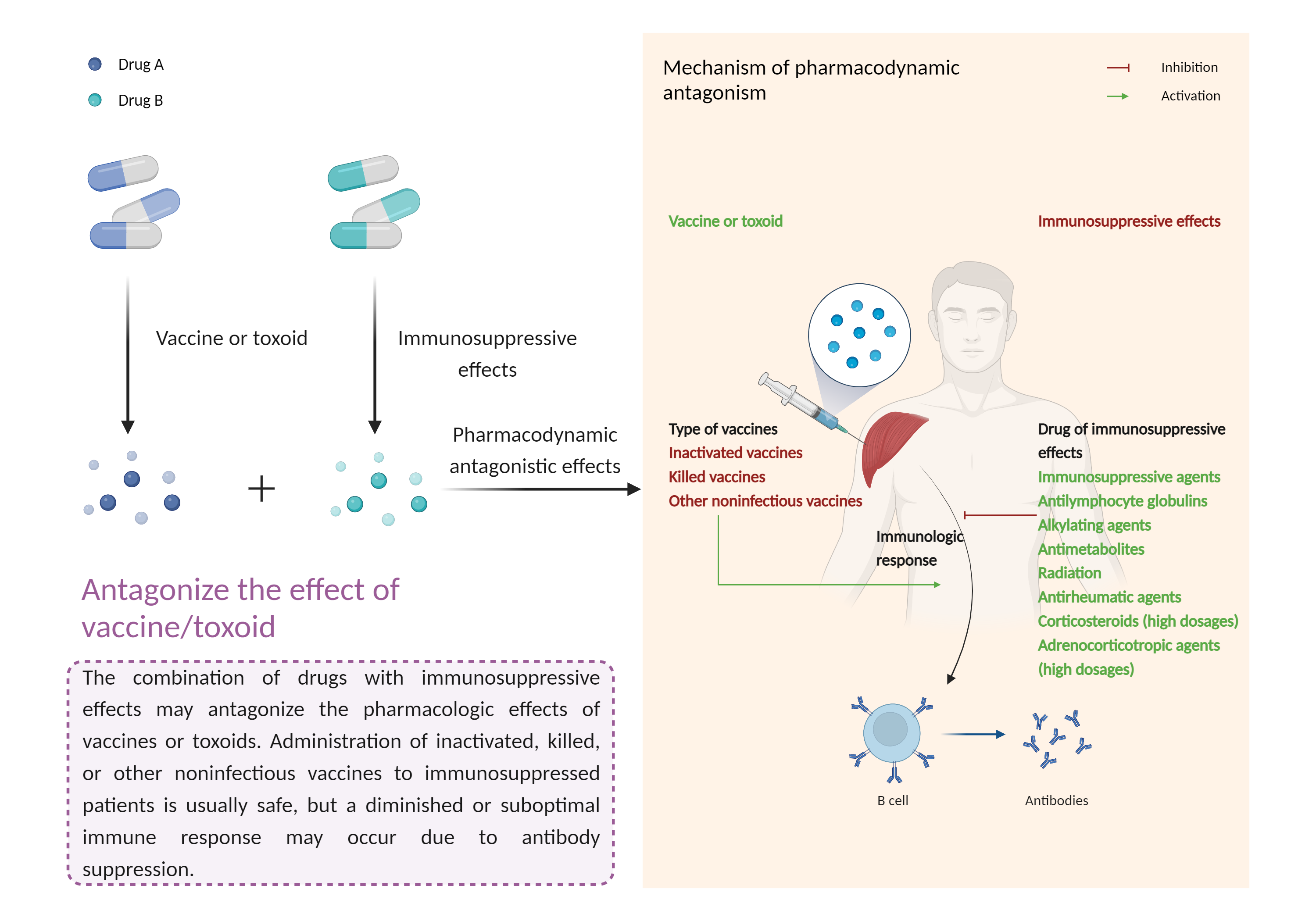
Routine nonemergency smallpox vaccination is considered a relative contraindication in patients receiving immunosuppressive corticosteroid therapy or ophthalmic corticosteroids. Vaccination should be deferred until after such therapy is discontinued and immune function has been restored. The interval depends on the dosage, duration, and route of corticosteroid therapy administered, but may be at least 3 months in most cases of systemic corticosteroid use. In patients who have recently been vaccinated, high-dose corticosteroid therapy should not be initiated for at least 2 weeks. Household contacts of immunosuppressed patients should also not be vaccinated. However, there are no absolute contraindications to vaccination if a high-risk exposure has occurred. In an outbreak emergency, smallpox vaccine is generally recommended for all persons, regardless of medical conditions. The risk for experiencing serious complications from the vaccine should be weighed against the risk of acquiring a potentially fatal smallpox infection. Vaccines may generally be administered to patients receiving corticosteroids as replacement therapy (e.g., for Addison's disease). |