Details of Drug-Drug Interaction
| Drug General Information (ID: DDILDB3X8C) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ipilimumab | Drug Info | Vemurafenib | Drug Info | |||||
| Drug Type | Monoclonal antibody | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Multikinase Inhibitors | |||||||
| Mechanism of Ipilimumab-Vemurafenib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of hepatotoxicity Click to Show/Hide Mechanism Graph | |||||||||
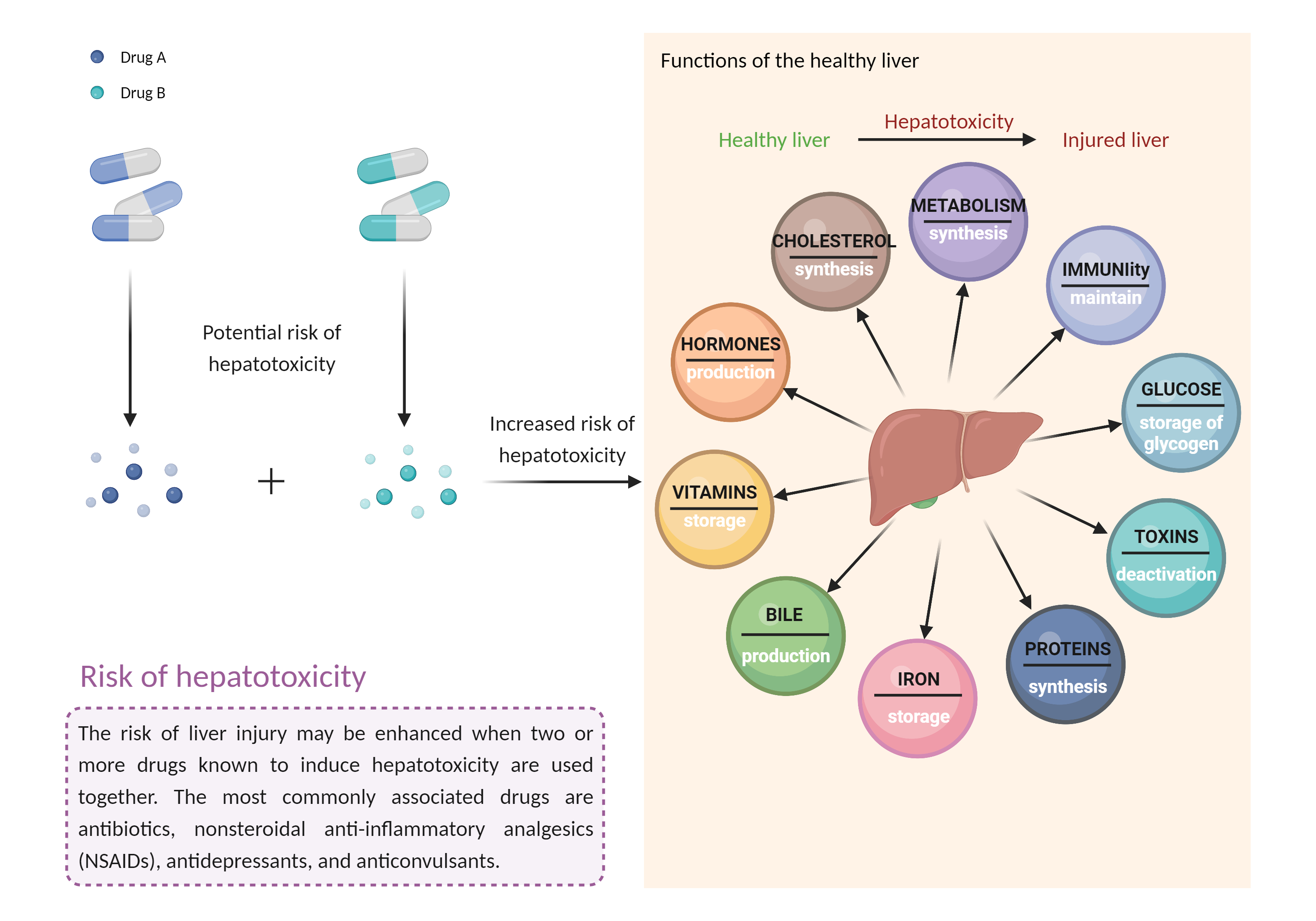 |
|||||||||
| Drug Name | Ipilimumab | Vemurafenib | |||||||
| Mechanism | Hepatotoxicity | Hepatotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hepatotoxicity | ||||||||
| Factor Description | Combination of drugs that can induce hepatotoxicity may increase the risk of liver injury. Symptoms vary depending on the level of exposure and the total extent of liver damage, and may cause few symptoms if the damage is mild, and eventually lead to liver failure in patients with severe damage. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The safety and effectiveness of vemurafenib in combination with ipilimumab have not been established. Liver transaminases, alkaline phosphatase, and bilirubin should be monitored prior to initiation of vemurafenib treatment and monthly during treatment, or as clinically indicated. Laboratory abnormalities should be managed with dosage reduction, treatment interruption, or treatment discontinuation in accordance with the product labeling. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Yervoy (ipilimumab). Bristol-Myers Squibb, Princeton, NJ. | ||||||||||||||||||
| 2 | Product Information. Zelboraf (vemurafenib). Genentech, South San Francisco, CA. | ||||||||||||||||||
