Details of Drug-Drug Interaction
| Drug General Information (ID: DDIL0A9ZTK) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Ioxilan | Drug Info | Tyropanoic acid | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Non-Ionic Iodinated Contrast Media | Diagnostic Radiopharmaceuticals | |||||||
| Structure | |||||||||
| Mechanism of Ioxilan-Tyropanoic acid Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
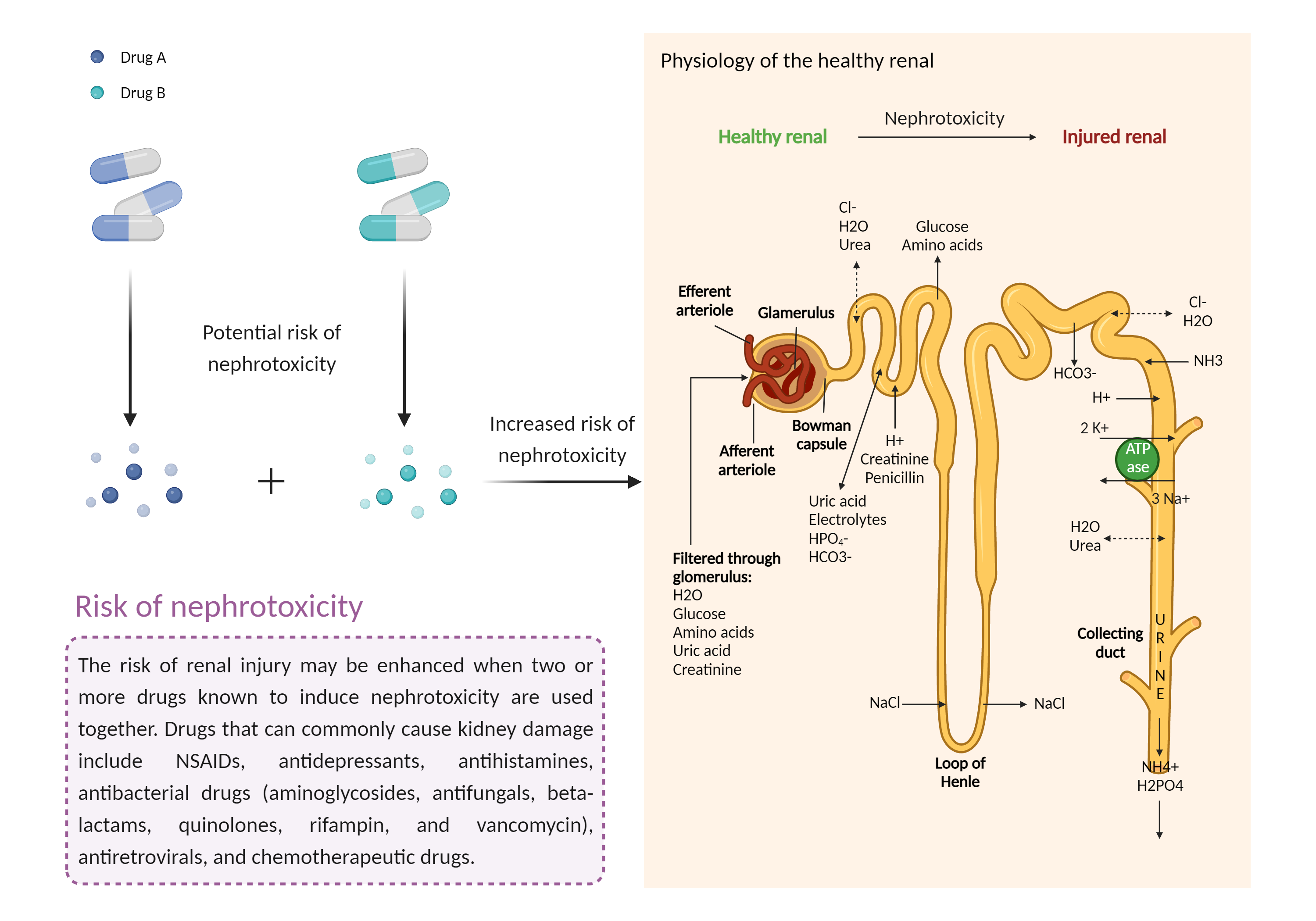
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Ioxilan | Tyropanoic acid | |||||||
| Mechanism | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Administration of any intravascular contrast agent should be deferred for 48 hours in patients with hepatic or biliary disorders who have recently received an oral cholecystographic contrast agent. | ||||||||
