Details of Drug-Drug Interaction
| Drug General Information (ID: DDIKERL5IA) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Salicylic acid (sodium) | Drug Info | Calcium carbonate | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Antacids/Minerals And Electrolytes | |||||||
| Structure | |||||||||
| Mechanism of Salicylic acid (sodium)-Calcium carbonate Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
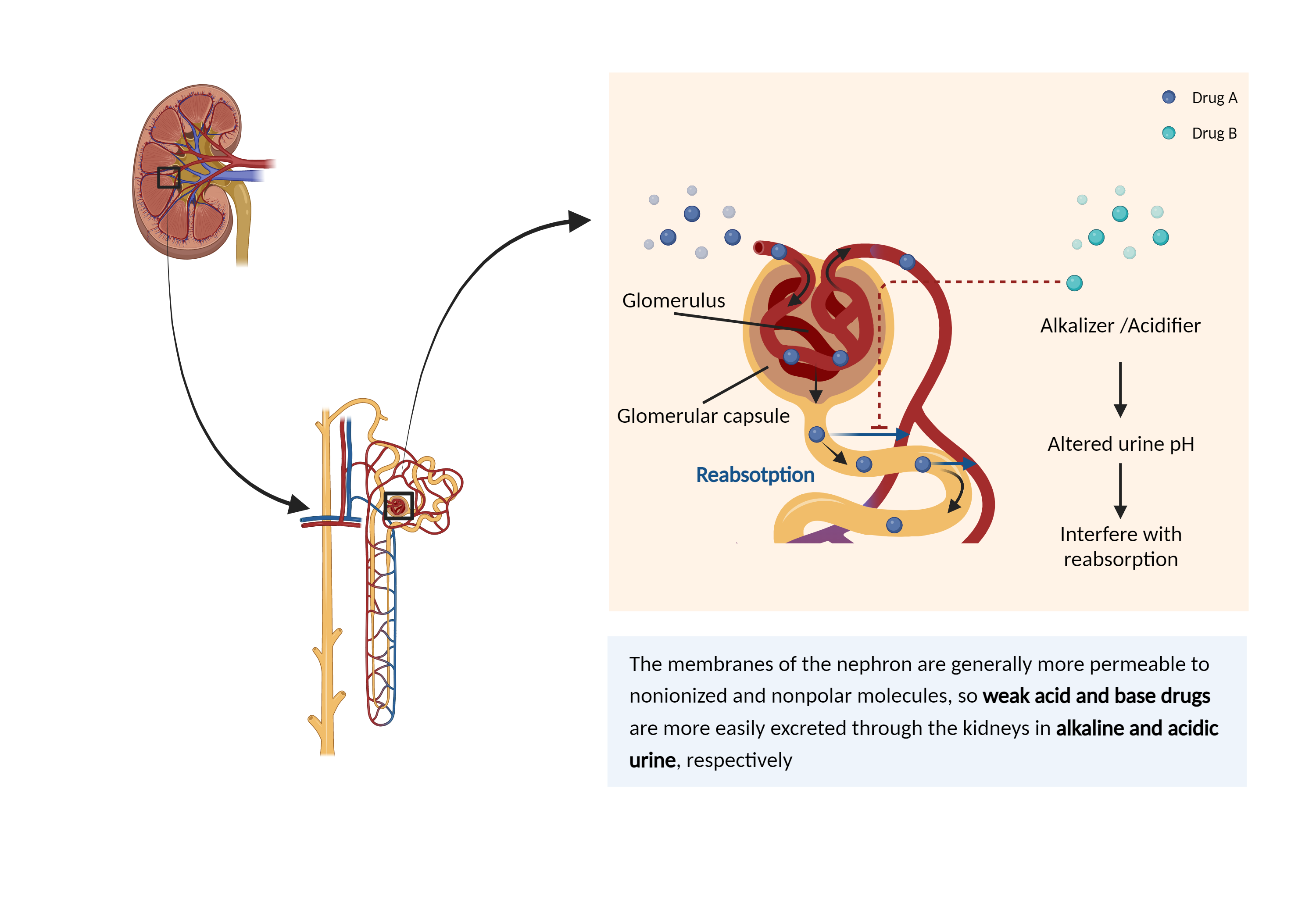
| Altered urine pH Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Salicylic acid (sodium) | Calcium carbonate | |||||||
| Mechanism | Urine pH sensitive | Urine alkalinizer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Urine pH | ||||||||
| Factor Description | The normal range of urine pH is between 4.5 and 8, depending mainly on the hydrogen ion concentration. Changes in urine pH may significantly affect drug reabsorption and excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Patients treated chronically with antacids (or oral medications that contain antacids such as didanosine buffered tablets or pediatric oral solution) and large doses of salicylates (i.e. 3 g/day or more) should be monitored for potentially diminished or inadequate analgesic and anti-inflammatory effects, and the salicylate dosage adjusted if necessary. | ||||||||
