Details of Drug-Drug Interaction
| Drug General Information (ID: DDIK07Q8PV) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Terbinafine | Drug Info | Valbenazine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antifungal Agents | Vmat2 Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Terbinafine-Valbenazine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
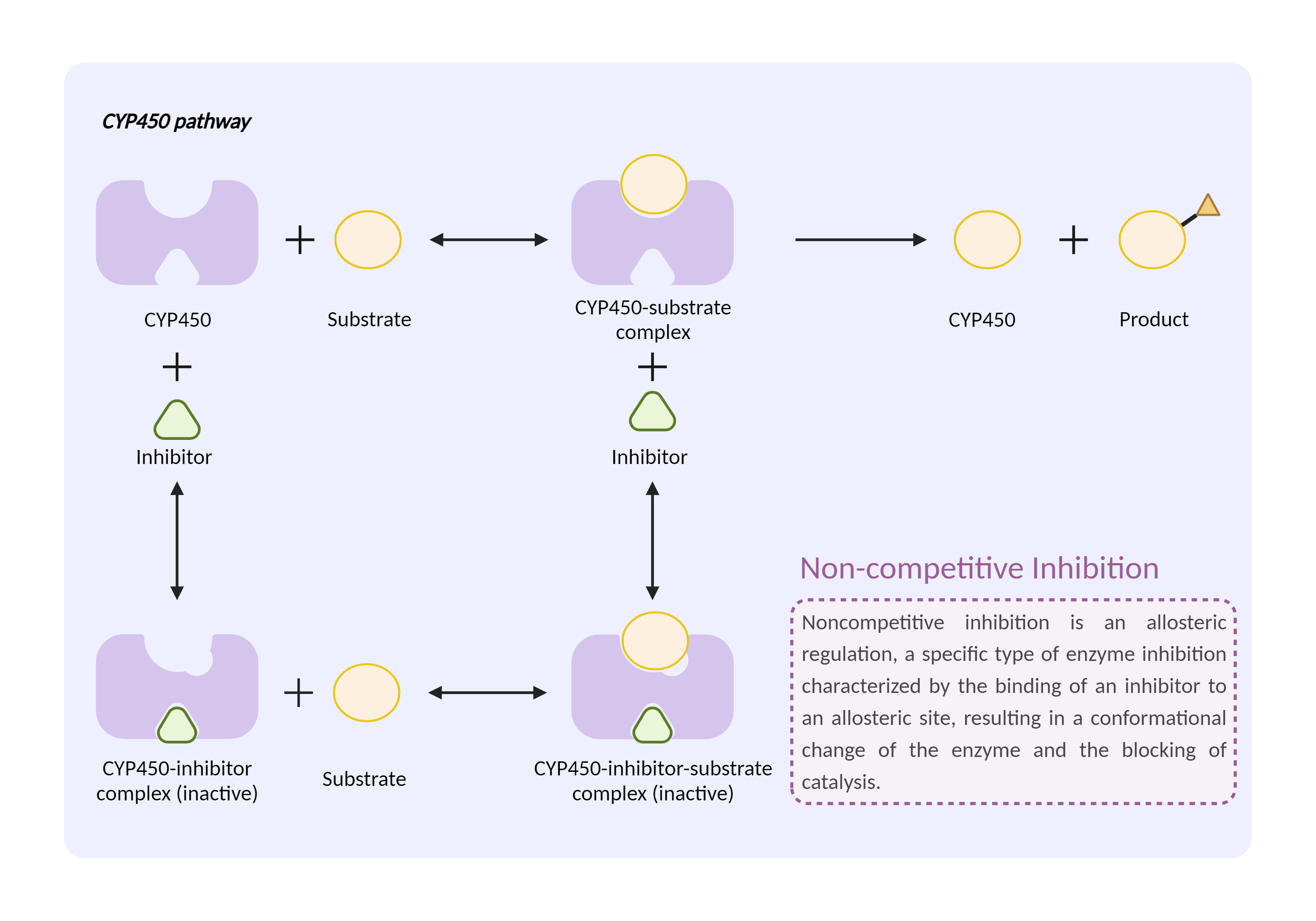 |
|||||||||
| Drug Name | Terbinafine | Valbenazine | |||||||
| Mechanism 1 | CYP450 2D6 inhibitor | CYP450 2D6 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2D6 |
×
Structure
Sequence
MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQLRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVFLARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDKAVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKVLRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVADLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVIHEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHFLDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGVFAFLVSPSPYELCAVPR
|
|||||||
| Gene Name | CYP2D6 | ||||||||
| Uniprot ID | CP2D6_HUMAN | ||||||||
| KEGG Pathway | hsa:1565 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of fatty acids, steroids and retinoids (PubMed:18698000, PubMed:19965576, PubMed:20972997, PubMed:21289075, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:18698000, PubMed:19965576, PubMed:20972997, PubMed:21289075, PubMed:21576599). Catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) (PubMed:19965576, PubMed:20972997). Metabolizes endocannabinoid arachidonoylethanolamide (anandamide) to 20-hydroxyeicosatetraenoic acid ethanolamide (20-HETE-EA) and 8,9-, 11,12-, and 14,15-epoxyeicosatrienoic acid ethanolamides (EpETrE-EAs), potentially modulating endocannabinoid system signaling (PubMed:18698000, PubMed:21289075). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). Catalyzes the oxidative transformations of all-trans retinol to all-trans retinal, a precursor for the active form all-trans-retinoic acid (PubMed:10681376). Also involved in the oxidative metabolism of drugs such as antiarrhythmics, adrenoceptor antagonists, and tricyclic antidepressants.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Additive CNS depression effects Click to Show/Hide Mechanism Graph | |||||||||
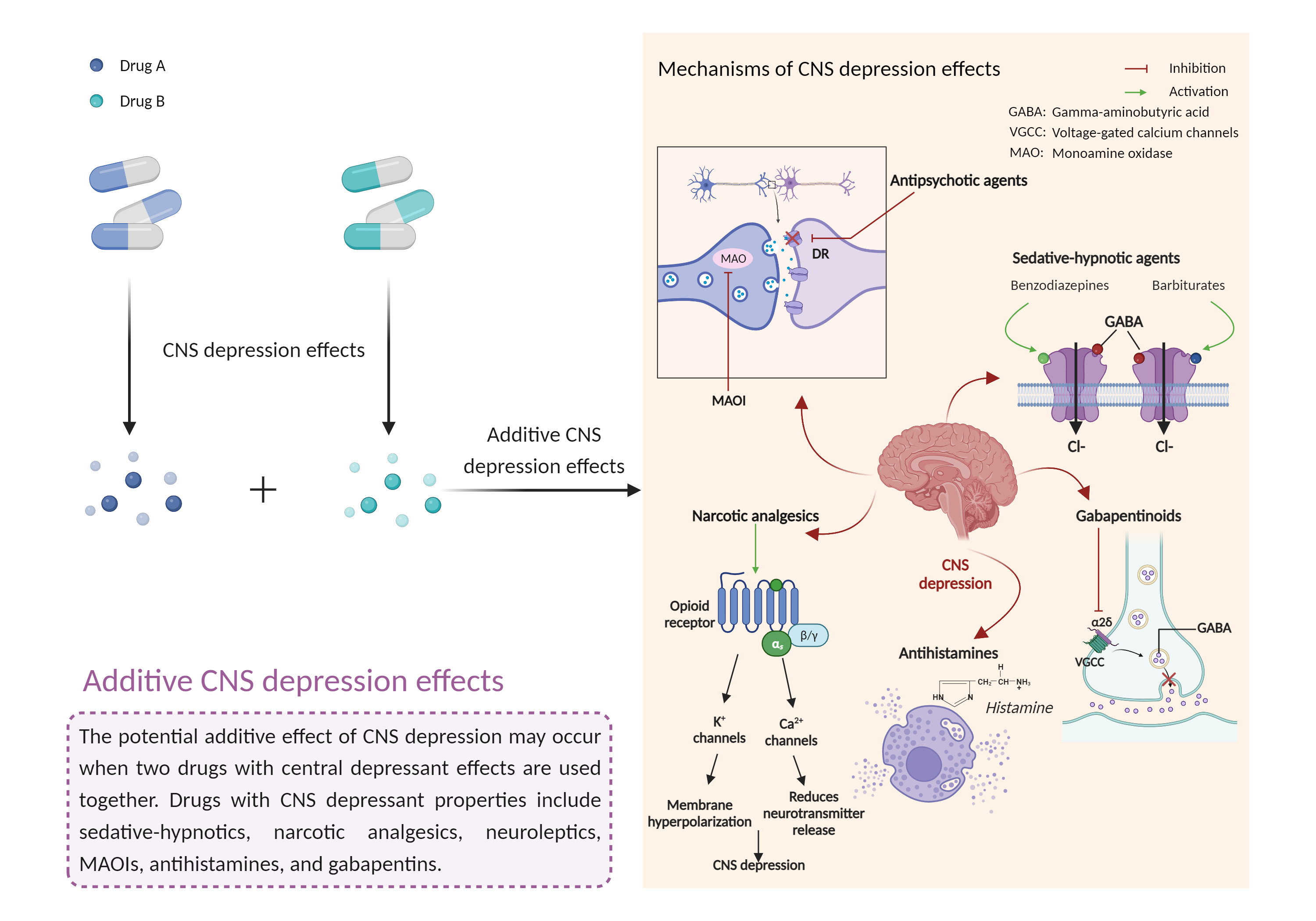 |
|||||||||
| Drug Name | Terbinafine | Valbenazine | |||||||
| Mechanism 2 | CNS depression effects | CNS depression effects | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | CNS depression effects | ||||||||
| Factor Description | CNS depressants are drugs that inhibit or suppress brain activity and can reduce mental and physical processes. Excessive CNS depression can lead to decreased heart rate, slow breathing (less than 10 breaths per minute), extreme confusion or loss of memory, nausea and vomiting, poor judgment, blue lips or fingertips, irritability and aggression, and clammy or cold skin. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Pharmacologic response to valbenazine should be monitored more closely whenever a strong inhibitor of CYP450 2D6 (e.g., paroxetine, fluoxetine, quinidine) is added to or withdrawn from therapy. Valbenazine is not recommended for use in patients with congenital long QT syndrome or with arrhythmias associated with a prolonged QT interval. Assessment of baseline QT interval and periodic monitoring during therapy may be considered. The manufacturer recommends a dosage reduction for valbenazine during concomitant administration with strong CYP450 2D6 inhibitors or in patients who are poor metabolizers of CYP450 2D6. Patients should be advised to seek prompt medical attention if they experience symptoms that could indicate the occurrence of torsade de pointes such as dizziness, lightheadedness, fainting, palpitation, irregular heart rhythm, shortness of breath, or syncope. When valbenazine is used in combination with other drugs that cause CNS depression, patients should be monitored for potentially excessive or prolonged CNS depression. Ambulatory patients should be counseled to avoid hazardous activities requiring mental alertness and motor coordination until they know how these agents affect them, and to notify their doctor if they experience excessive or prolonged CNS effects that interfere with their normal activities. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Ingrezza (valbenazine). Neurocrine Biosciences, Inc., San Diego, CA. | ||||||||||||||||||
