Details of Drug-Drug Interaction
| Drug General Information (ID: DDIJ8CYUIB) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Fluvoxamine | Drug Info | Theophylline | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidepressants | Bronchodilators | |||||||
| Structure | |||||||||
| Mechanism of Fluvoxamine-Theophylline Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
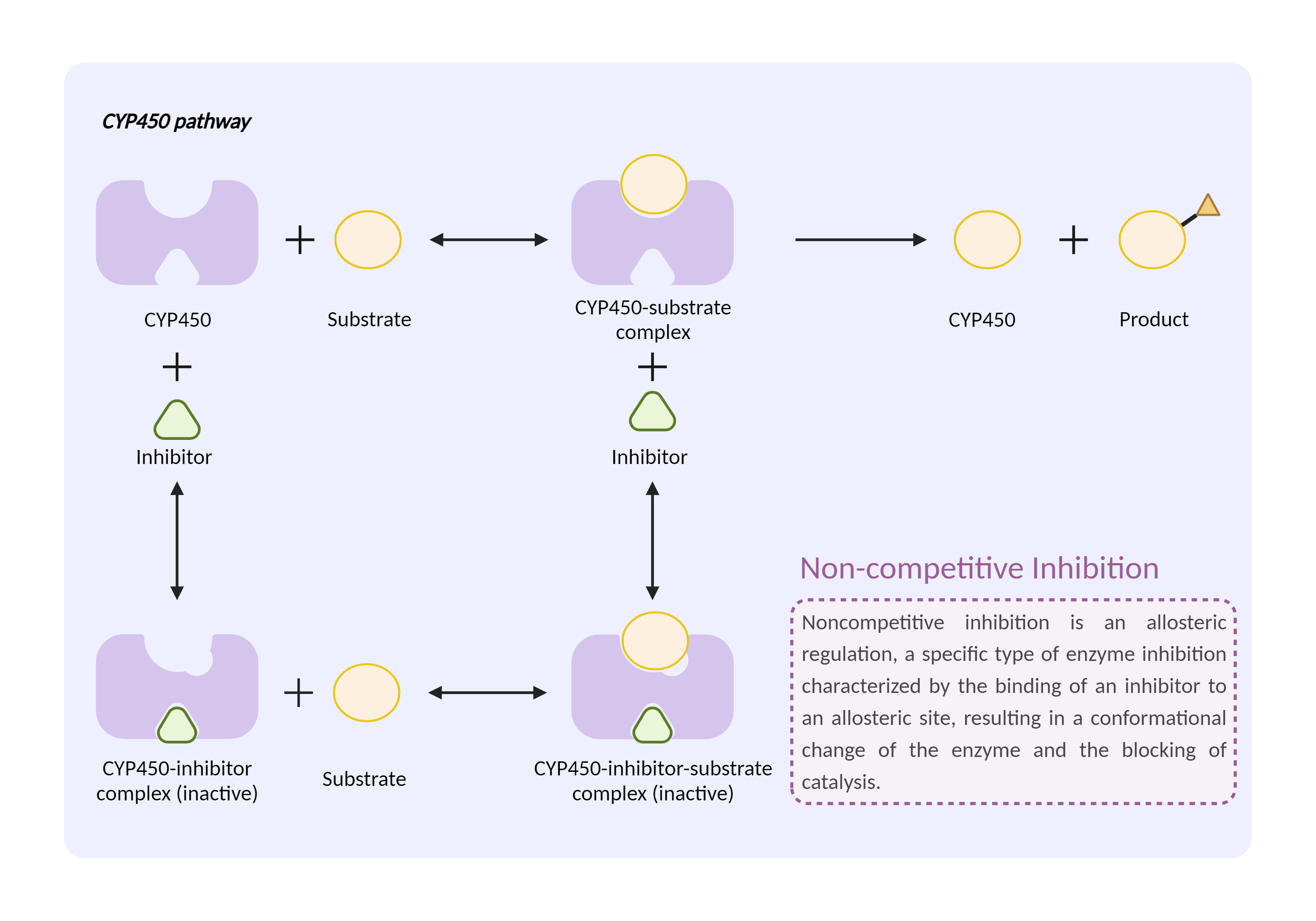 |
|||||||||
| Drug Name | Fluvoxamine | Theophylline | |||||||
| Mechanism | CYP450 1A2 inhibitor | CYP450 1A2 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 1A2 |
×
Structure
Sequence
MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKNPHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDGQSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELMAGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFPILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGNLIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLSDRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPELWEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLEFSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|||||||
| Gene Name | CYP1A2 | ||||||||
| Uniprot ID | CP1A2_HUMAN | ||||||||
| KEGG Pathway | hsa:1544 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids, steroid hormones and vitamins (PubMed:9435160, PubMed:10681376, PubMed:11555828, PubMed:12865317, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:9435160, PubMed:10681376, PubMed:11555828, PubMed:12865317, PubMed:19965576). Catalyzes the hydroxylation of carbon-hydrogen bonds (PubMed:11555828, PubMed:12865317). Exhibits high catalytic activity for the formation of hydroxyestrogens from estrone (E1) and 17beta-estradiol (E2), namely 2-hydroxy E1 and E2 (PubMed:11555828, PubMed:12865317). Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). May act as a major enzyme for all-trans retinoic acid biosynthesis in the liver. Catalyzes two successive oxidative transformation of all-trans retinol to all-trans retinal and then to the active form all-trans retinoic acid (PubMed:10681376). Primarily catalyzes stereoselective epoxidation of the last double bond of polyunsaturated fatty acids (PUFA), displaying a strong preference for the (R,S) stereoisomer (PubMed:19965576). Catalyzes bisallylic hydroxylation and omega-1 hydroxylation of PUFA (PubMed:9435160). May also participate in eicosanoids metabolism by converting hydroperoxide species into oxo metabolites (lipoxygenase-like reaction, NADPH-independent) (PubMed:21068195). Plays a role in the oxidative metabolism of xenobiotics. Catalyzes the N-hydroxylation of heterocyclic amines and the O-deethylation of phenacetin (PubMed:14725854). Metabolizes caffeine via N3-demethylation (Probable).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The use of theophylline or its salts in combination with fluvoxamine should generally be avoided. If coadministration is required, a reduction of theophylline dosage by one-half to two-thirds should be considered. Pharmacologic response and serum levels should be closely monitored following initiation, discontinuation or change of dosage of fluvoxamine, and the theophylline dosage adjusted accordingly. Patients should be advised to contact their physician if they experience signs and symptoms suggestive of theophylline toxicity such as nausea, vomiting, diarrhea, anorexia, headache, tremor, irritability, confusion, insomnia, seizure, palpitation, and arrhythmia. Other selective serotonin reuptake inhibitors including citalopram, escitalopram, fluoxetine, paroxetine, and sertraline do not significantly inhibit CYP450 1A2 and may be safer alternatives in theophylline-treated patients. | ||||||||
