Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIWTBKHQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Lansoprazole | Drug Info | Tolevamer | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Proton Pump Inhibitors | Potassium-Binding Resin | |||||||
| Structure | |||||||||
| Mechanism of Lansoprazole-Tolevamer Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
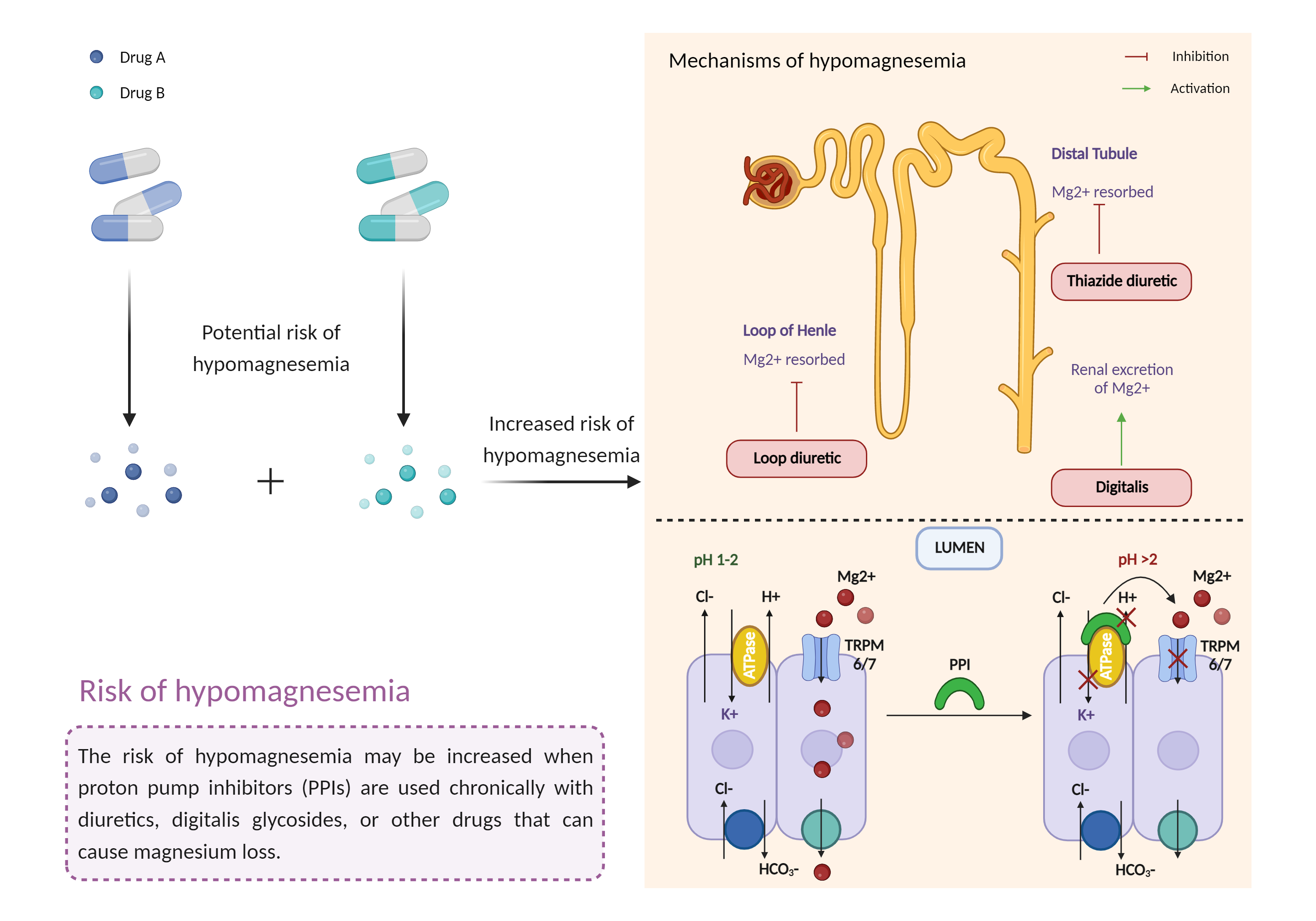
| Increased risk of hypomagnesemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Lansoprazole | Tolevamer | |||||||
| Mechanism | Hypomagnesemia | Hypomagnesemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hypomagnesemia | ||||||||
| Factor Description | Hypomagnesemia is a condition that occurs when you have too much magnesium in your body. Symptoms of hypomagnesemia include: nausea, vomiting, neurological damage, abnormally low blood pressure (hypotension), flushing, and headaches. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Monitoring of serum magnesium levels is recommended prior to initiation of therapy and periodically thereafter if prolonged treatment with a proton pump inhibitor is anticipated or when combined with other agents that can cause hypomagnesemia such as diuretics, aminoglycosides, cation exchange resins, amphotericin B, cetuximab, cisplatin, cyclosporine, foscarnet, panitumumab, pentamidine, and tacrolimus. Patients should be advised to seek immediate medical attention if they develop potential signs and symptoms of hypomagnesemia such as palpitations, arrhythmia, muscle spasm, tremor, or convulsions. In children, abnormal heart rates may cause fatigue, upset stomach, dizziness, and lightheadedness. Magnesium replacement as well as discontinuation of the PPI may be required in some patients. | ||||||||
