Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIR9DOAS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nabumetone | Drug Info | Pemetrexed | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Nsaids/Analgesics | Antineoplastics | |||||||
| Structure | |||||||||
| Mechanism of Nabumetone-Pemetrexed Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
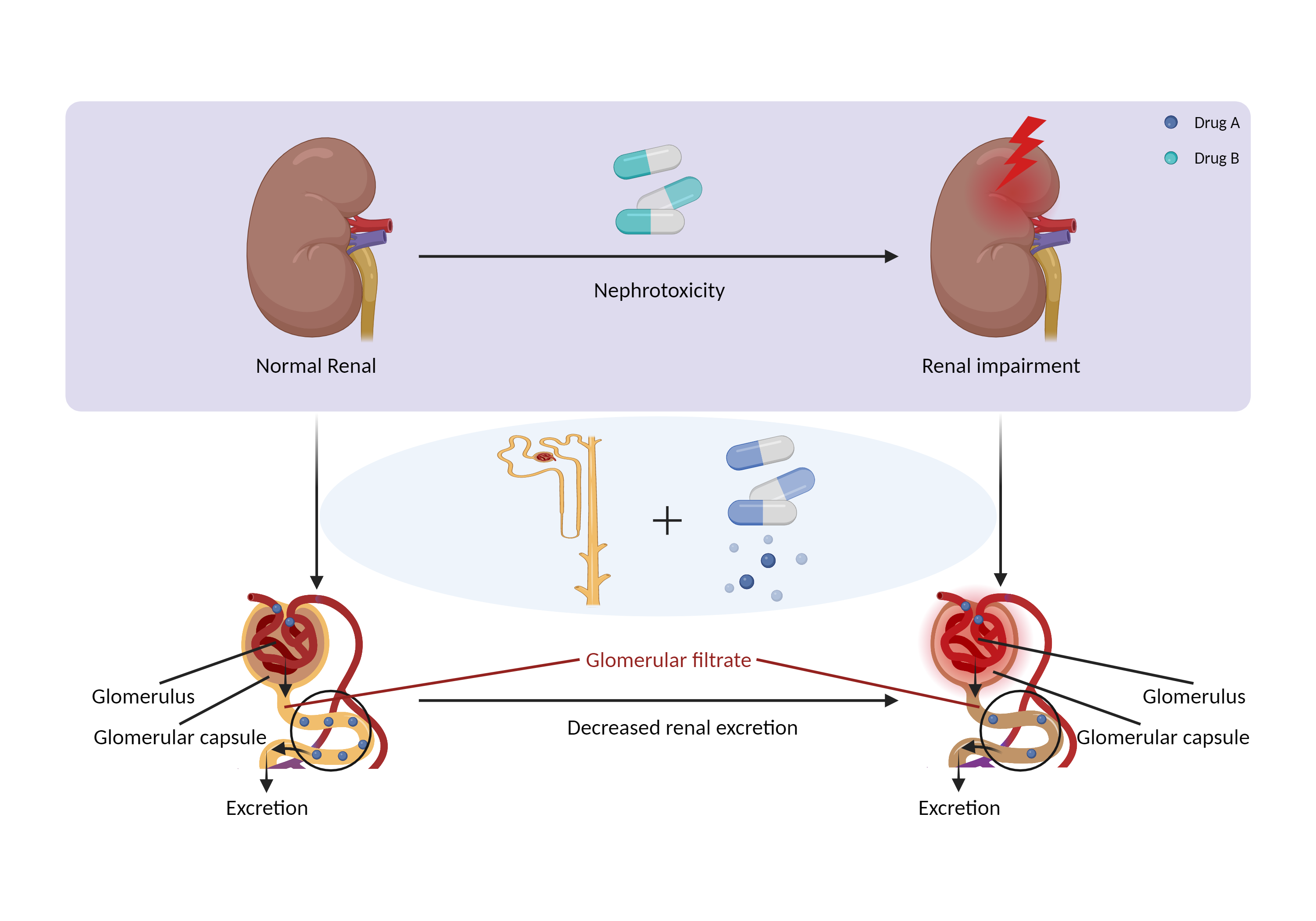
| Decreased renal excretion due to nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Nabumetone | Pemetrexed | |||||||
| Mechanism 1 | Nephrotoxicity | Primarily eliminated by renal excretion | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | Drug-induced nephrotoxicity is a common and potentially serious complication of medication use. The kidneys are the primary organ for drug excretion, and when they are damaged can lead to decreased drug excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 |
Nephrotoxicity Decrease renal perfusion |
Primarily eliminated by renal excretion | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | Drug-induced nephrotoxicity is a common and potentially serious complication of medication use. The kidneys are the primary organ for drug excretion, and when they are damaged can lead to decreased drug excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Mechanism 3 | Decrease renal perfusion/blood flow rate | Primarily eliminated by renal excretion | |||||||
| Key Mechanism Factor 3 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | Drug-induced nephrotoxicity is a common and potentially serious complication of medication use. The kidneys are the primary organ for drug excretion, and when they are damaged can lead to decreased drug excretion. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Ibuprofen at 400 mg and aspirin at 325 mg four times a day, or less, may be used with pemetrexed in patients with normal renal function. However, caution is advised in patients with mild to moderate renal insufficiency (creatinine clearance 45 to 79 mL/min). These patients should avoid taking NSAIDs with short elimination half-lives (e.g., diclofenac, etodolac, fenoprofen, flurbiprofen, ibuprofen, indomethacin, ketoprofen, ketorolac, meclofenamate, mefenamic acid, sulindac, tolmetin, low dosages of salicylates) 2 days before to 2 days after pemetrexed administration. If concomitant administration is necessary, patients should be monitored closely for toxicity, especially myelosuppression, nephrotoxicity, and gastrointestinal toxicity. | ||||||||
