Details of Drug-Drug Interaction
| Drug General Information (ID: DDIIASR1XJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Liotrix | Drug Info | Simethicone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Thyroid Drugs | Gi Agents | |||||||
| Structure | |||||||||
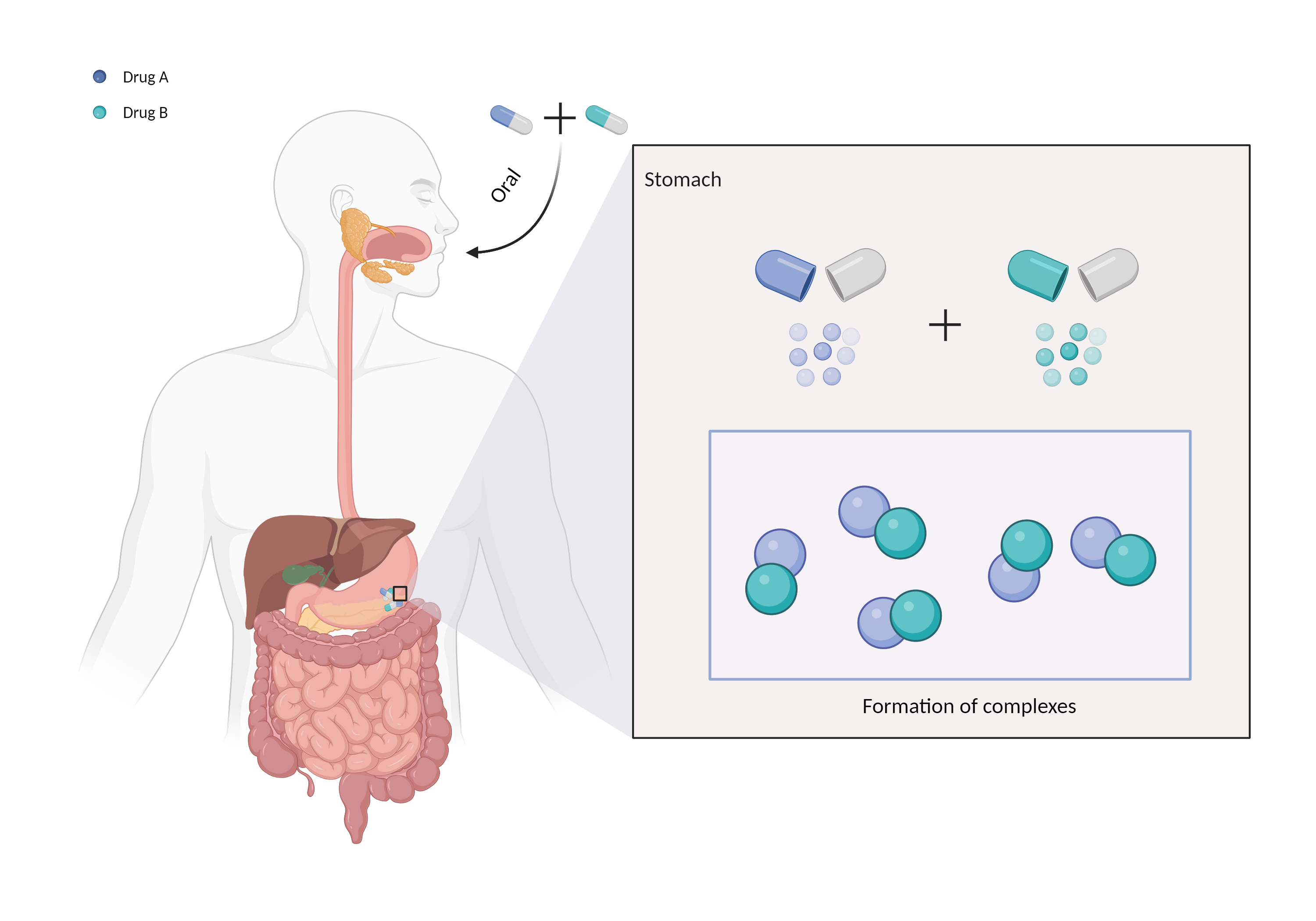
| Mechanism of Liotrix-Simethicone Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Complex formation Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Liotrix | Simethicone | |||||||
| Mechanism | Binds to polyvalent cations | Polyvalent cations | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Chelation | ||||||||
| Factor Description | Chelation is a direct drug interaction that usually involves the formation of dimers or trimers, resulting in larger complexes and poorer absorption. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | It is recommended to separate the times of administration of levothyroxine and simethicone and antacids or other antacid-containing preparations (e.g., didanosine buffered tablets or pediatric oral solution) by at least 4 hours. Monitoring of serum TSH levels is recommended. Patients with gastrointestinal or malabsorption disorders may be at a greater risk of developing clinical or subclinical hypothyroidism due to this interaction. | ||||||||
