Details of Drug-Drug Interaction
| Drug General Information (ID: DDIHS25VGJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Drotrecogin alfa | Drug Info | Betrixaban | Drug Info | |||||
| Drug Type | Protein/peptide | Small molecule | |||||||
| Therapeutic Class | Coagulation Modifiers | Anticoagulants/Factor Xa Inhibitors | |||||||
| Mechanism of Drotrecogin alfa-Betrixaban Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
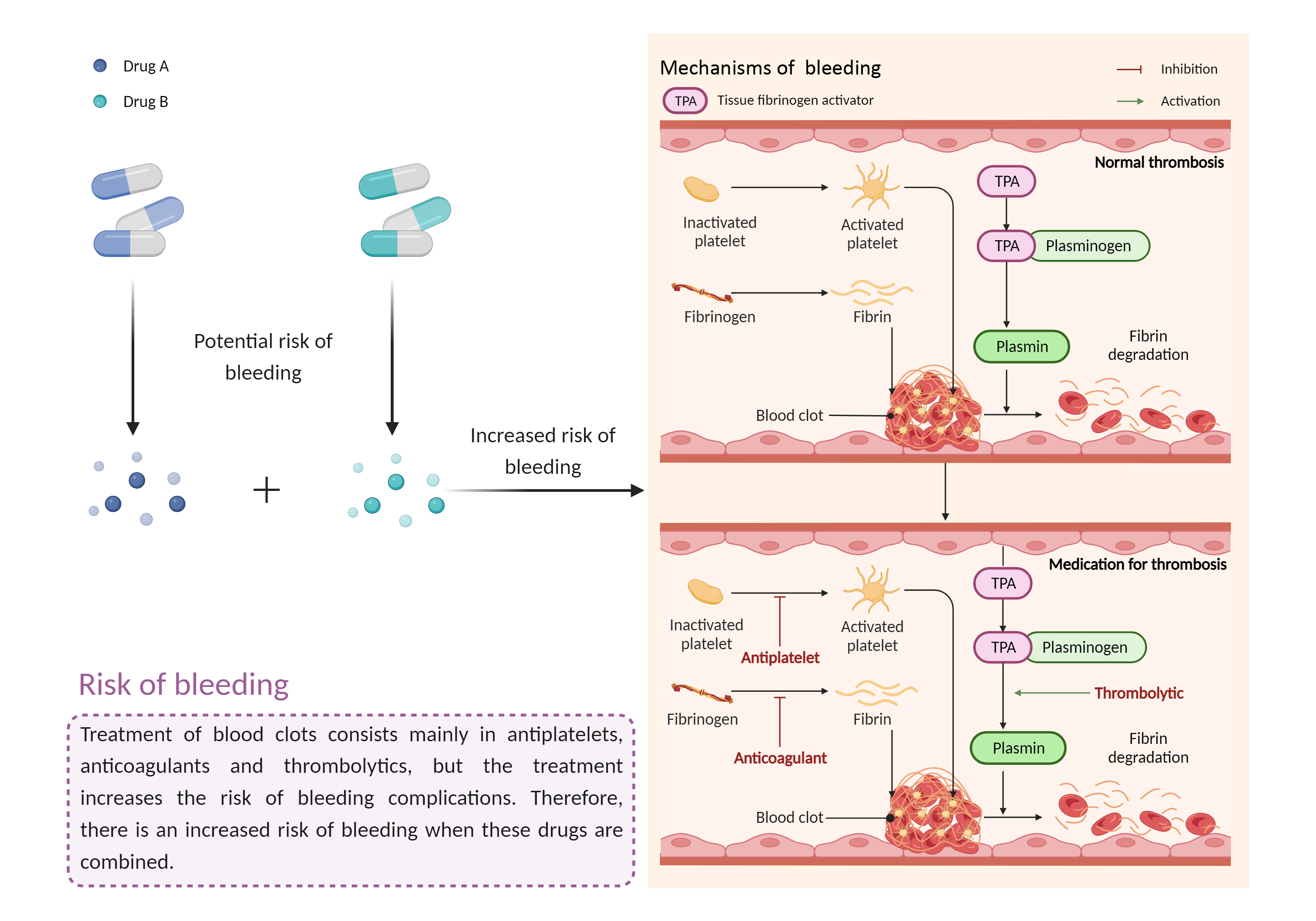 |
|||||||||
| Drug Name | Drotrecogin alfa | Betrixaban | |||||||
| Mechanism |
Risk of bleeding Anticoagulant effects |
Risk of bleeding Anticoagulant |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The potentially increased risk of bleeding versus the benefits of drotrecogin alfa therapy should be carefully considered in seriously ill septic patients who have recently (within 7 days) received oral anticoagulants, platelet inhibitors, or aspirin (more than 650 mg/day). Close clinical and laboratory monitoring for bleeding complications is recommended if concurrent therapy is required. Drotrecogin alfa should be discontinued immediately if clinically significant bleeding occurs. Continued use of other agents that may have contributed to the bleeding should be carefully assessed. Once adequate hemostasis is attained, drotrecogin alfa may be resumed if necessary. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Xigris (drotrecogin alfa). Lilly, Eli and Company, Indianapolis, IN. | ||||||||||||||||||
