Details of Drug-Drug Interaction
| Drug General Information (ID: DDIH2ZFXO3) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Moexipril | Drug Info | Sacubitril | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antihypertensive Agents | Neprilysin Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Moexipril-Sacubitril Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
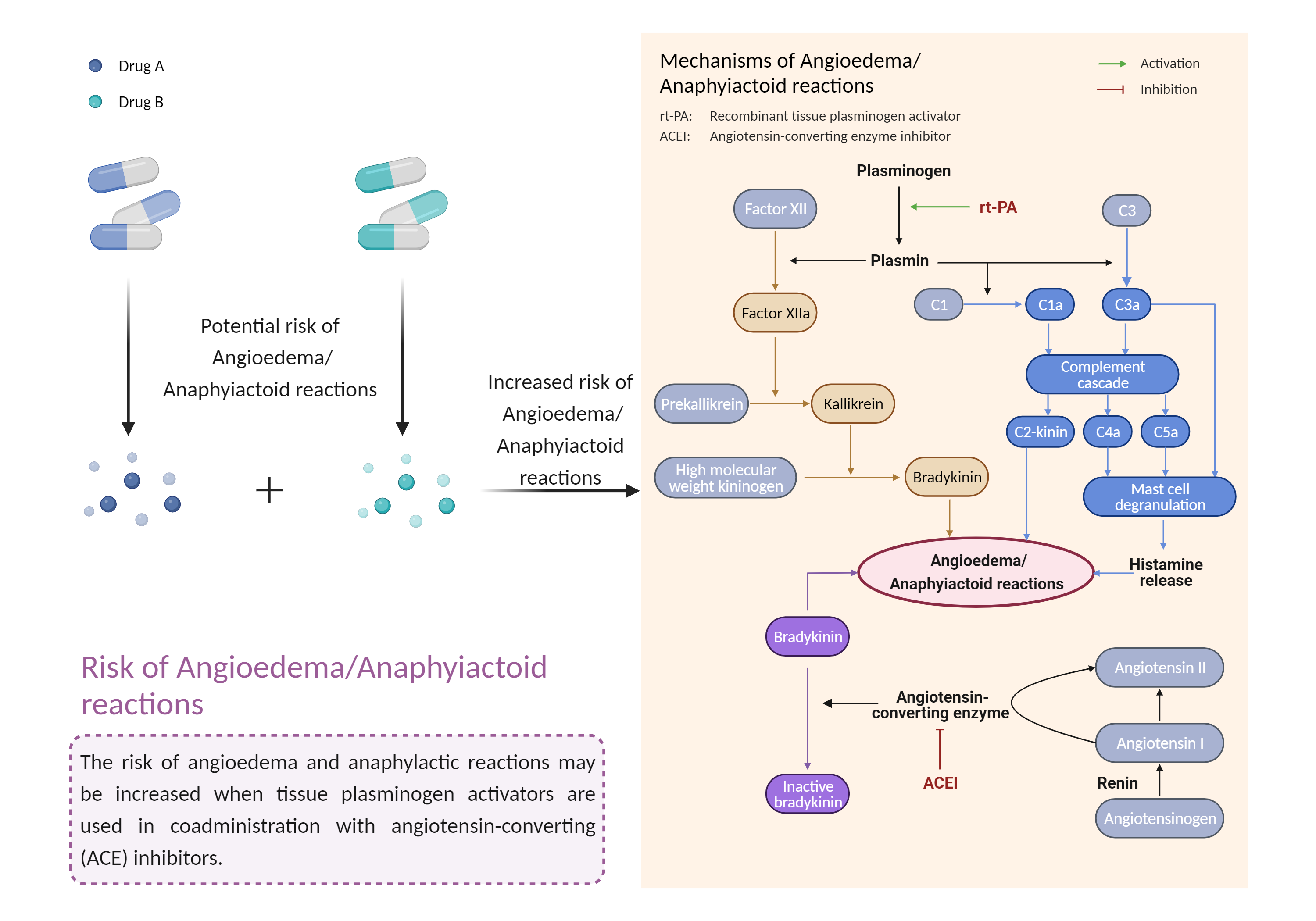
| Increased risk of angioedema/anaphylactoid reactions Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Moexipril | Sacubitril | |||||||
| Mechanism | Angioedema | Angioedema | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Angioedema/anaphylactoid reactions | ||||||||
| Factor Description | Anaphylactoid reaction is a condition in which symptoms similar to an allergic reaction occur, but no immune sensitization is detected. Most commonly, the syndrome begins with skin involvement, usually pruritus, flushing, urticaria, or angioedema. Angioedema is a reaction to a trigger that causes swelling of the tissue below the inner layer of the skin, called the dermis or submucosa, and the swelling can occur on the face, throat, or around the genitals. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Dual blockade of the RAA system by adding an ACE inhibitor to an angiotensin II receptor antagonist (i.e., valsartan) is considered contraindicated. Use of sacubitril-valsartan or an ACE inhibitor within 36 hours of each other should be avoided. | ||||||||
