Details of Drug-Drug Interaction
| Drug General Information (ID: DDIGST0QHU) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Diclofenamide | Drug Info | Eslicarbazepine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Ophthalmic Glaucoma Agents | Anticonvulsants | |||||||
| Structure | |||||||||
| Mechanism of Diclofenamide-Eslicarbazepine Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
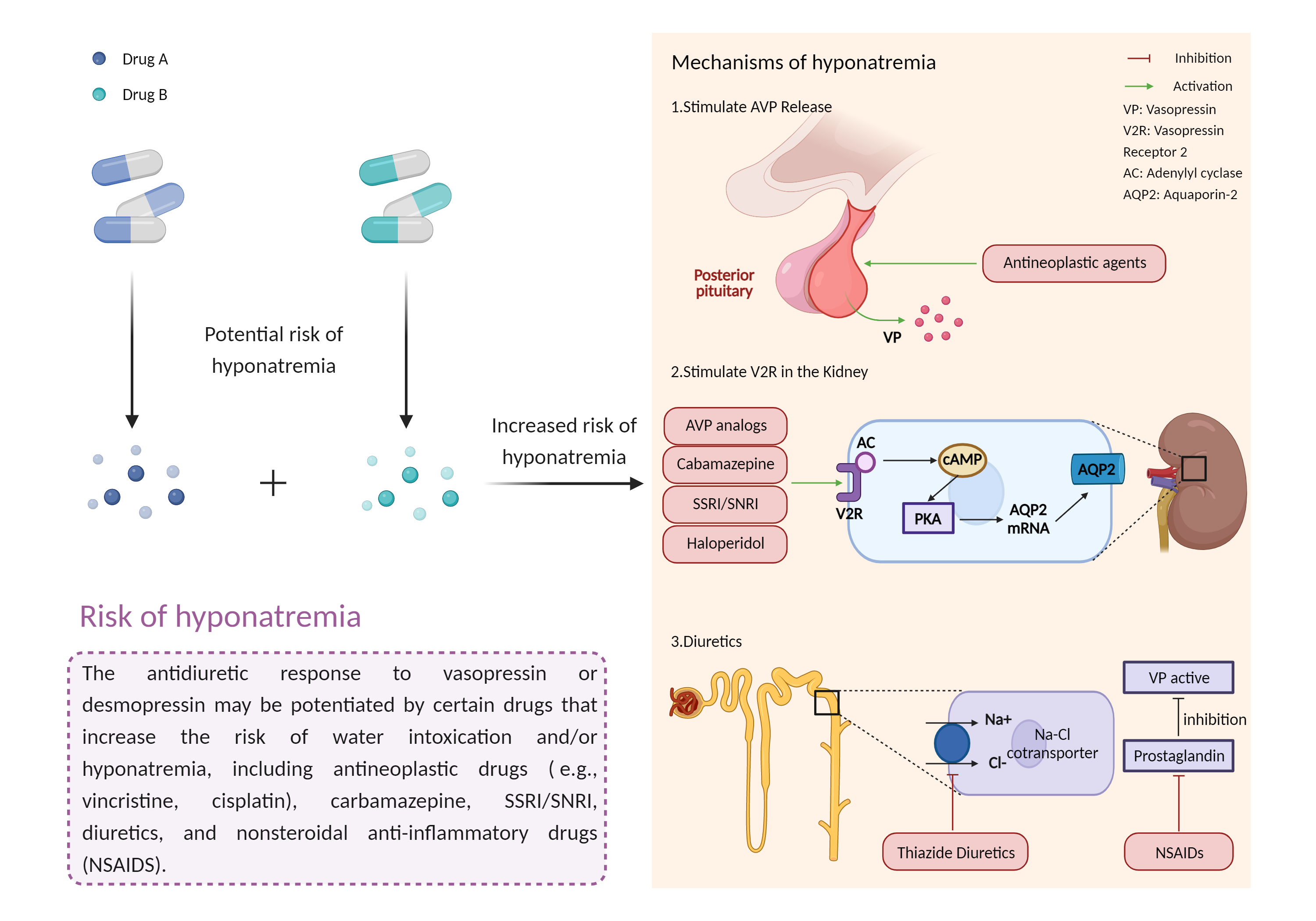
| Increased risk of hyponatremia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Diclofenamide | Eslicarbazepine | |||||||
| Mechanism | Hyponatremia | Hyponatremia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hyponatremia | ||||||||
| Factor Description | Hyponatremia is a condition in which the amount of sodium in the blood is lower than normal. Symptoms of hyponatremia may include: nausea and vomiting, fatigue, headache or confusion, muscle cramps, weakness, irritability and restlessness. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Routine monitoring of serum sodium and chloride levels should be considered during maintenance treatment with eslicarbazepine acetate and concomitant use of other medications known to decrease serum sodium levels. Patients should be advised to seek medical attention if they experience potential signs and symptoms of hyponatremia such as nausea, vomiting, headache, malaise, lethargy, irritability, difficulty concentrating, memory impairment, confusion, weakness, muscle spasm, and unsteadiness (which may lead to falls). More severe and/or acute cases may include hallucination, syncope, seizure, coma, respiratory arrest, and death. Depending on the severity of hyponatremia, dosage reduction or discontinuation of eslicarbazepine acetate and other medications may be required, and appropriate medical intervention instituted as necessary. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Aptiom (eslicarbazepine). Sunovion Pharmaceuticals Inc, Marlborough, MA. | ||||||||||||||||||
