Details of Drug-Drug Interaction
| Drug General Information (ID: DDIGDNUYO8) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Nateglinide | Drug Info | Sulfinpyrazone | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antidiabetic Agents | Uricosuric Agents | |||||||
| Structure | |||||||||
| Mechanism of Nateglinide-Sulfinpyrazone Interaction (Severity Level: Minor) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme inhibition Click to Show/Hide Mechanism Graph | |||||||||
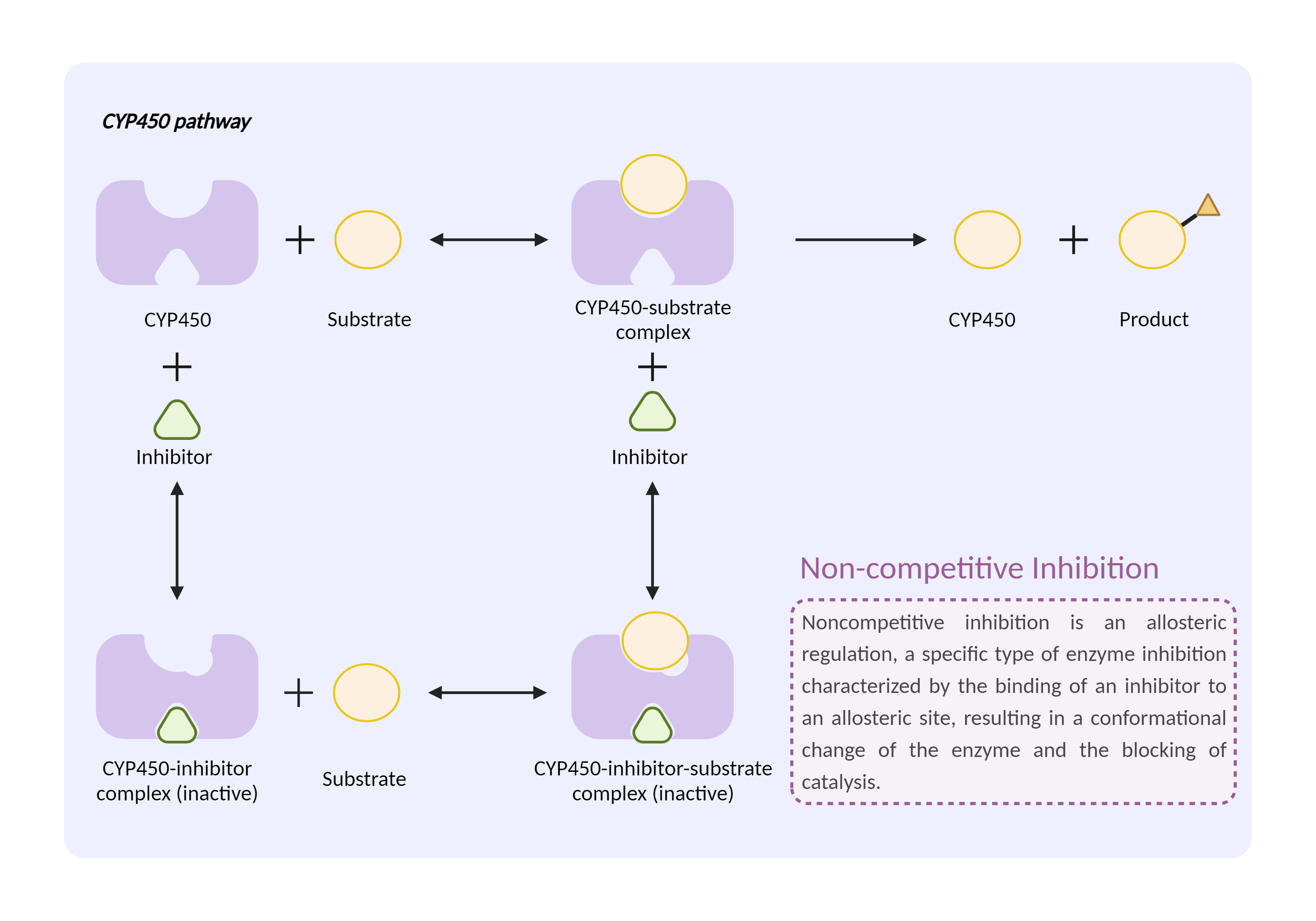 |
|||||||||
| Drug Name | Nateglinide | Sulfinpyrazone | |||||||
| Mechanism | CYP450 2C9 substrate | CYP450 2C9 inhibitor | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2C9 |
×
Structure
Sequence
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
|
|||||||
| Gene Name | CYP2C9 | ||||||||
| Uniprot ID | CP2C9_HUMAN | ||||||||
| KEGG Pathway | hsa:1559 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of various endogenous substrates, including fatty acids and steroids (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:7574697, PubMed:9866708, PubMed:9435160, PubMed:12865317, PubMed:15766564, PubMed:19965576, PubMed:21576599). Catalyzes the epoxidation of double bonds of polyunsaturated fatty acids (PUFA) (PubMed:7574697, PubMed:15766564, PubMed:19965576, PubMed:9866708). Catalyzes the hydroxylation of carbon-hydrogen bonds. Metabolizes cholesterol toward 25-hydroxycholesterol, a physiological regulator of cellular cholesterol homeostasis (PubMed:21576599). Exhibits low catalytic activity for the formation of catechol estrogens from 17beta-estradiol (E2) and estrone (E1), namely 2-hydroxy E1 and E2 (PubMed:12865317). Catalyzes bisallylic hydroxylation and hydroxylation with double-bond migration of polyunsaturated fatty acids (PUFA) (PubMed:9866708, PubMed:9435160). Also metabolizes plant monoterpenes such as limonene. Oxygenates (R)- and (S)-limonene to produce carveol and perillyl alcohol (PubMed:11950794). Contributes to the wide pharmacokinetics variability of the metabolism of drugs such as S-warfarin, diclofenac, phenytoin, tolbutamide and losartan (PubMed:25994031).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
