Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFW17TPS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Carfilzomib | Drug Info | Norgestrel | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Protease Inhibitors | Contraceptive Agents | |||||||
| Structure | |||||||||
| Mechanism of Carfilzomib-Norgestrel Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
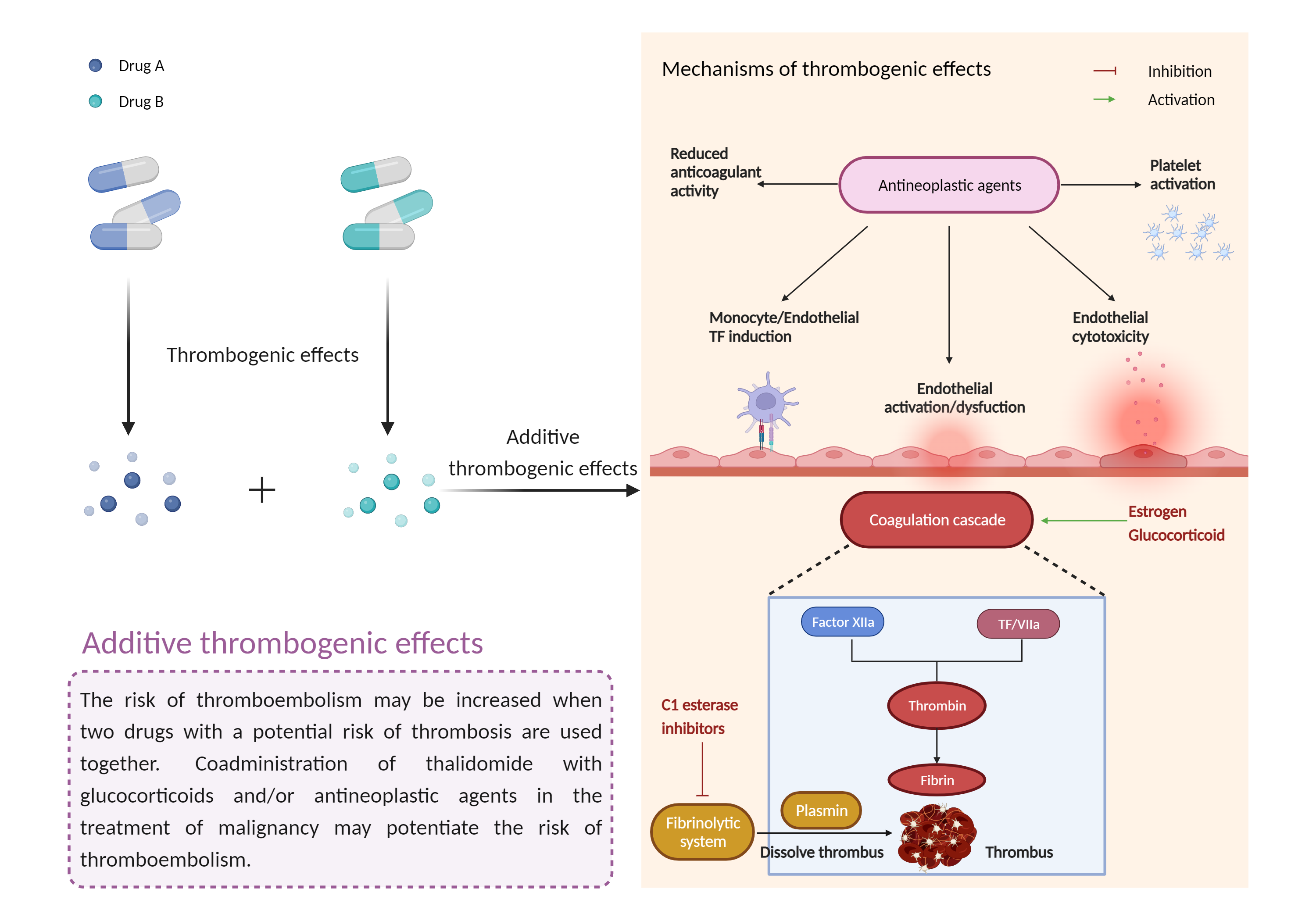
| Additive thrombogenic effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Carfilzomib | Norgestrel | |||||||
| Mechanism | Thrombogenic effects | Thrombogenic effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Thrombogenic effects | ||||||||
| Factor Description | Drug-induced thrombosis disrupts the balance between thrombogenic factors and protective mechanisms and tends to produce thrombosis. Drug-induced thrombosis usually causes venous thrombotic events, and arterial thrombotic events are also noted as a result of drug administration | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Hormonal contraception associated with a risk of thrombosis should generally be avoided during use of carfilzomib. Advise female and male patients of reproductive potential to use effective contraception or abstain from sexual activity during treatment with carfilzomib for at least 30 and 90 days, respectively, following completion of therapy. Advise the patient to contact their physician immediately if pregnancy does occur during these times. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Faculty of Sexual & Reproductive Healthcare "FSRH Clinical Guidance: Drug Interactions with Hormonal Contraception.". | ||||||||||||||||||
