Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFTO4LE7) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Argatroban | Drug Info | Edoxaban | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anticoagulants | Anticoagulants/Factor Xa Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Argatroban-Edoxaban Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
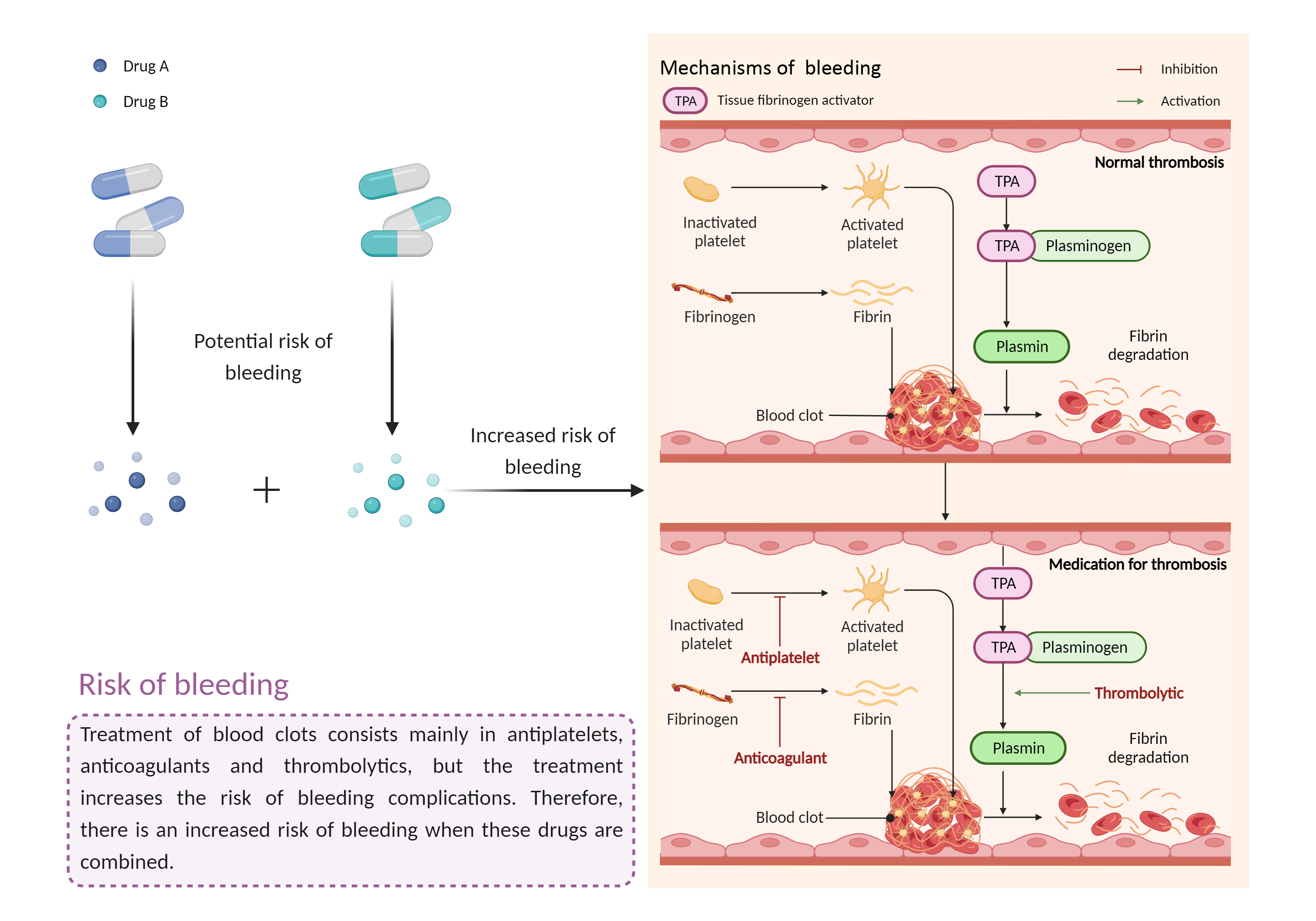
| Increased risk of bleeding Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Argatroban | Edoxaban | |||||||
| Mechanism |
Risk of bleeding Anticoagulant |
Risk of bleeding Anticoagulant |
|||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Bleeding | ||||||||
| Factor Description | Patients may be at risk for bleeding when the outcome of a disease or disease treatment confounds the standard mechanisms for maintaining hemostasis. Signs or symptoms of abnormal bleeding include: bleeding that takes a long time to stop (including nosebleeds, bleeding gums, bleeding from cuts and abrasions, and menstrual bleeding); severe unexplained bruising, or bruising that becomes larger; blood in the urine or stool, etc. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Due to increased bleeding risk, the use of edoxaban with other anticoagulants should generally be avoided except during therapeutic transition periods where patients should be observed closely. Patients should be routinely evaluated for signs and symptoms suggesting blood loss such as a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress (in pregnant women). In patients undergoing neuraxial intervention, the use of edoxaban should be approached with caution and only after thorough assessment of risks and benefits. Besides bleeding complications, patients should also be monitored frequently for signs and symptoms of neurologic impairment such as midline back pain, sensory and motor deficits (numbness or weakness in lower limbs), and bowel or bladder dysfunction. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Savaysa (edoxaban). Daiichi Sankyo, Inc., Parsippany, NJ. | ||||||||||||||||||
