Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFS6UOJC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Mefenamic acid | Drug Info | Binimetinib | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiinflammatory Agents | Multikinase Inhibitors | |||||||
| Structure | |||||||||
| Mechanism of Mefenamic acid-Binimetinib Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UGT inhibition Click to Show/Hide Mechanism Graph | |||||||||
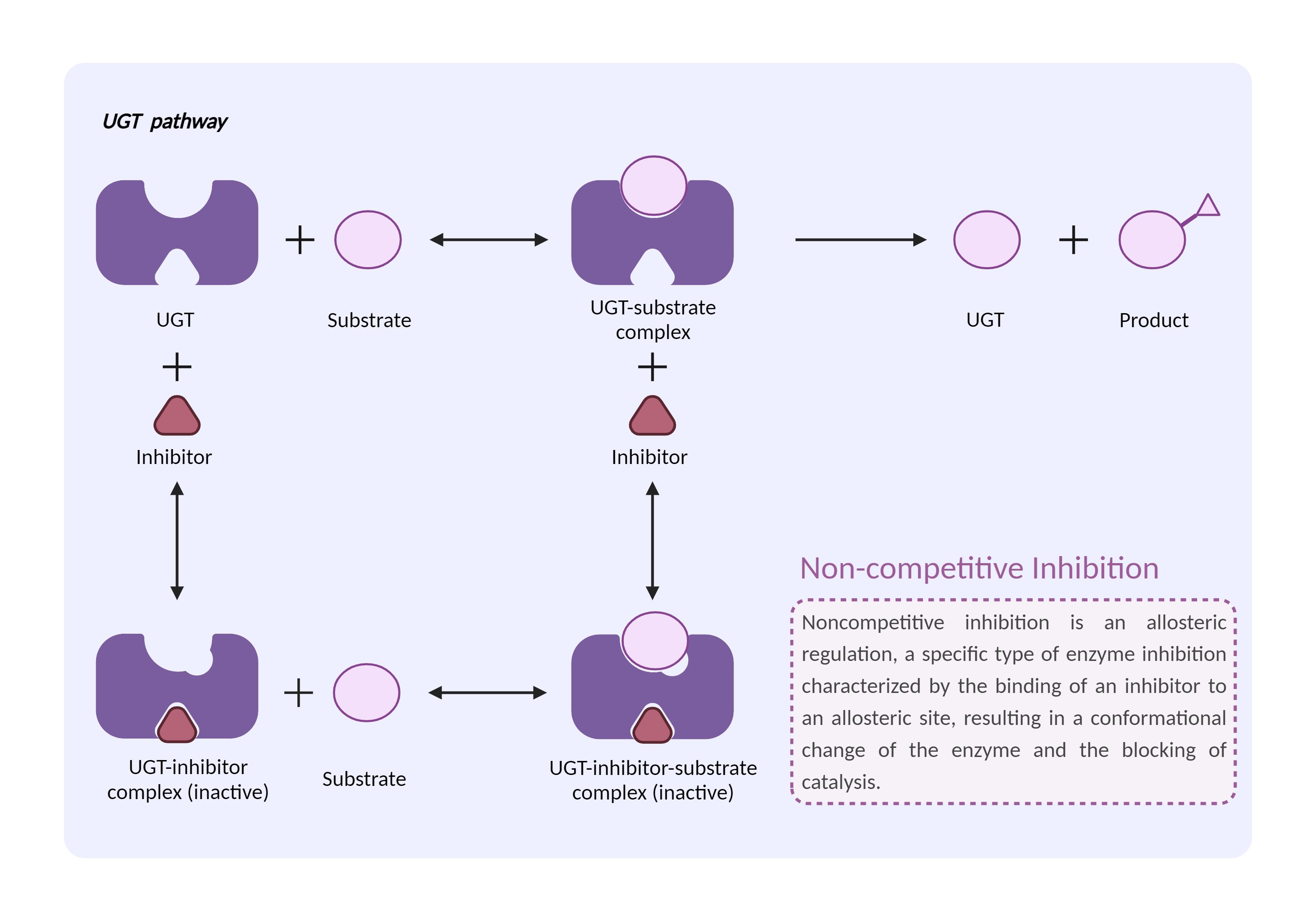 |
|||||||||
| Drug Name | Mefenamic acid | Binimetinib | |||||||
| Mechanism | UGT2B7 inhibitor | UGT2B7 substrate | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | UDP-glucuronosyltransferase 2B7 |
×
Structure
Sequence
MSVKWTSVILLIQLSFCFSSGNCGKVLVWAAEYSHWMNIKTILDELIQRGHEVTVLASSASILFDPNNSSALKIEIYPTSLTKTELENFIMQQIKRWSDLPKDTFWLYFSQVQEIMSIFGDITRKFCKDVVSNKKFMKKVQESRFDVIFADAIFPCSELLAELFNIPFVYSLSFSPGYTFEKHSGGFIFPPSYVPVVMSELTDQMTFMERVKNMIYVLYFDFWFEIFDMKKWDQFYSEVLGRPTTLSETMGKADVWLIRNSWNFQFPYPLLPNVDFVGGLHCKPAKPLPKEMEDFVQSSGENGVVVFSLGSMVSNMTEERANVIASALAQIPQKVLWRFDGNKPDTLGLNTRLYKWIPQNDLLGHPKTRAFITHGGANGIYEAIYHGIPMVGIPLFADQPDNIAHMKARGAAVRVDFNTMSSTDLLNALKRVINDPSYKENVMKLSRIQHDQPVKPLDRAVFWIEFVMRHKGAKHLRVAAHDLTWFQYHSLDVIGFLLVCVATVIFIVTKCCLFCFWKFARKAKKGKND
|
|||||||
| Gene Name | UGT2B7 | ||||||||
| Uniprot ID | UD2B7_HUMAN | ||||||||
| KEGG Pathway | hsa:7364 | ||||||||
| Protein Family | UDP-glycosyltransferase family | ||||||||
| Protein Function |
UDP-glucuronosyltransferase (UGT) that catalyzes phase II biotransformation reactions in which lipophilic substrates are conjugated with glucuronic acid to increase the metabolite's water solubility, thereby facilitating excretion into either the urine or bile (PubMed:10702251, PubMed:15472229, PubMed:15470161, PubMed:18674515, PubMed:18719240, PubMed:19022937, PubMed:23288867, PubMed:23756265, PubMed:26220143, PubMed:17442341). Essential for the elimination and detoxification of drugs, xenobiotics and endogenous compounds (PubMed:15470161, PubMed:18674515, PubMed:23756265). Catalyzes the glucuronidation of endogenous steroid hormones such as androgens (epitestosterone, androsterone) and estrogens (estradiol, epiestradiol, estriol, catechol estrogens) (PubMed:2159463, PubMed:15472229, PubMed:18719240, PubMed:19022937, PubMed:23288867, PubMed:26220143, PubMed:17442341). Also regulates the levels of retinoic acid, a major metabolite of vitamin A involved in apoptosis, cellular growth and differentiation, and embryonic development (PubMed:10702251). Contributes to bile acid (BA) detoxification by catalyzing the glucuronidation of BA substrates, which are natural detergents for dietary lipids absorption (PubMed:23756265). Involved in the glucuronidation of the AGTR1 angiotensin receptor antagonist losartan, caderastan and zolarsatan, drugs which can inhibit the effect of angiotensin II (PubMed:18674515). Also metabolizes mycophenolate, an immunosuppressive agent (PubMed:15470161).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Until further information is available, caution is recommended if binimetinib must be used concomitantly with P-gp, UGT 1A1, UGT 2B7, and/or BCRP inhibitors. Binimetinib should be monitored more closely whenever a P-gp, UGT 1A1, UGT 2B7, and/or BCRP inhibitor is added to or withdrawn from therapy, and the binimetinib dosage adjusted as necessary. | ||||||||
