Details of Drug-Drug Interaction
| Drug General Information (ID: DDIFPKMEVS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Amphotericin B (lipid complex) | Drug Info | Givosiran | Drug Info | |||||
| Drug Type | Small molecule | Small interfering RNA | |||||||
| Therapeutic Class | Antifungal Agents | Metabolic Agents | |||||||
| Structure | |||||||||
| Mechanism of Amphotericin B (lipid complex)-Givosiran Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
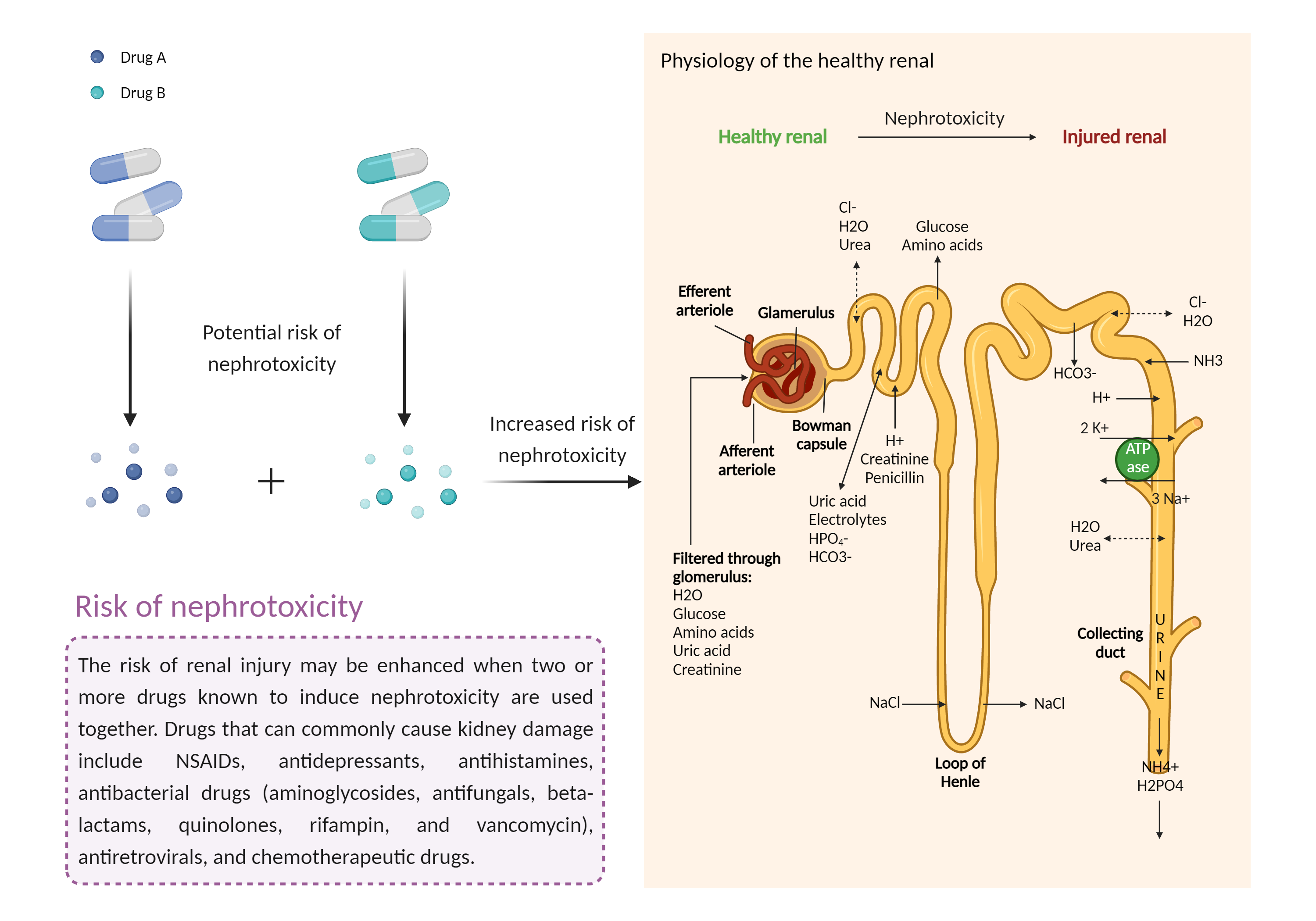
| Increased risk of nephrotoxicity Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Amphotericin B (lipid complex) | Givosiran | |||||||
| Mechanism | Nephrotoxicity | Nephrotoxicity | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Nephrotoxicity | ||||||||
| Factor Description | The combination of drugs that can induce nephrotoxicity may increase the risk of kidney injury. When kidney injury occurs, the inability to remove excess urine and waste from the body can lead to high levels of urea nitrogen, creatinine, and electrolytes (such as potassium and magnesium) in the blood. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Close clinical and laboratory monitoring of renal and neurologic function and drug serum concentrations (if clinically appropriate) is recommended. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "Australian Product Information.". | ||||||||||||||||||
| 2 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
