Details of Drug-Drug Interaction
| Drug General Information (ID: DDIE2A84PJ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Methoxyflurane | Drug Info | Rifampicin | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Anesthetics | Antituberculosis Agents | |||||||
| Structure | |||||||||
| Mechanism of Methoxyflurane-Rifampicin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| CYP450 enzyme induction Click to Show/Hide Mechanism Graph | |||||||||
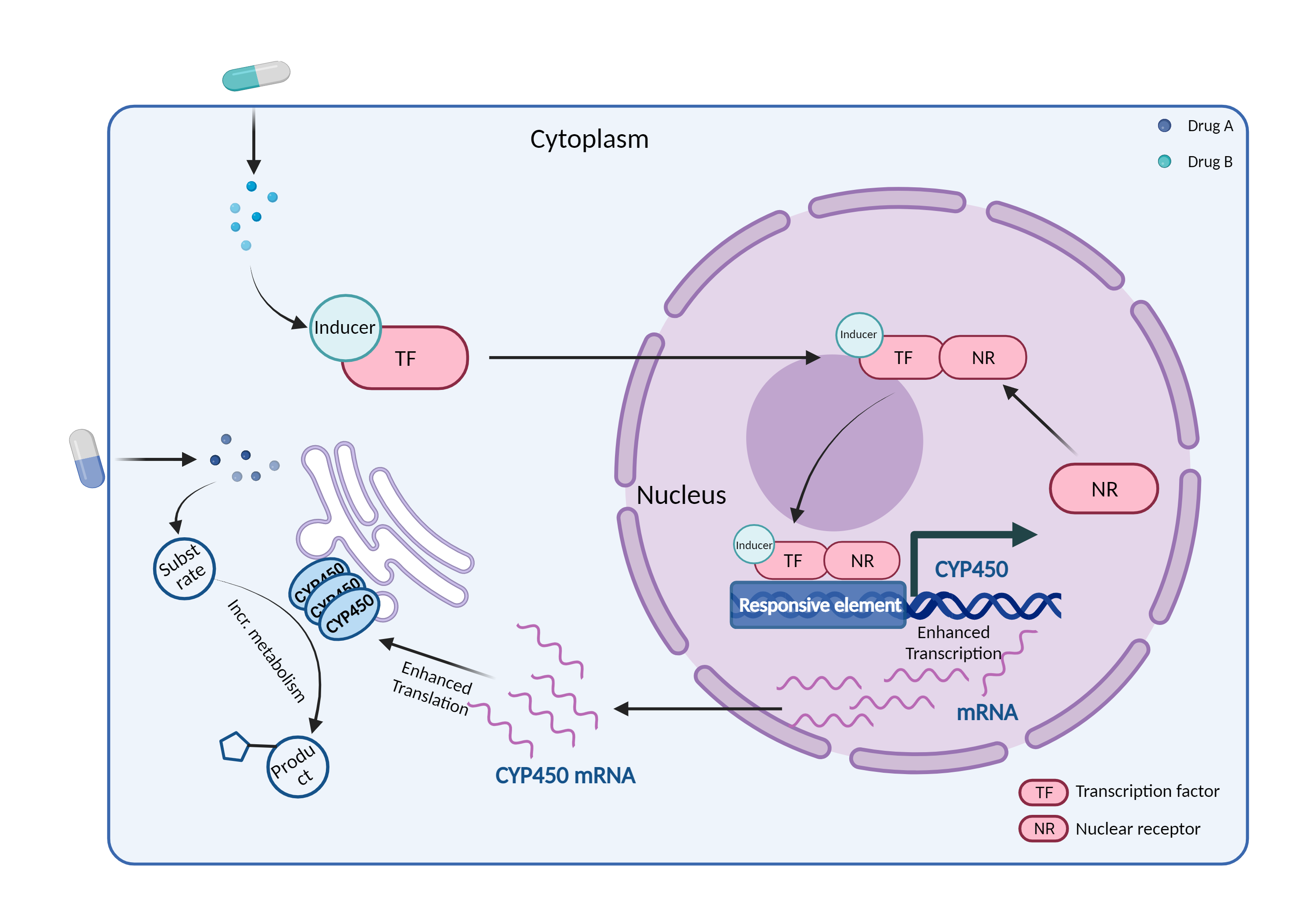 |
|||||||||
| Drug Name | Methoxyflurane | Rifampicin | |||||||
| Mechanism 1 | CYP450 2A6 substrate | CYP450 2A6 inducer | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Cytochrome P450 2A6 |
×
Structure
Sequence
MLASGMLLVALLVCLTVMVLMSVWQQRKSKGKLPPGPTPLPFIGNYLQLNTEQMYNSLMKISERYGPVFTIHLGPRRVVVLCGHDAVREALVDQAEEFSGRGEQATFDWVFKGYGVVFSNGERAKQLRRFSIATLRDFGVGKRGIEERIQEEAGFLIDALRGTGGANIDPTFFLSRTVSNVISSIVFGDRFDYKDKEFLSLLRMMLGIFQFTSTSTGQLYEMFSSVMKHLPGPQQQAFQLLQGLEDFIAKKVEHNQRTLDPNSPRDFIDSFLIRMQEEEKNPNTEFYLKNLVMTTLNLFIGGTETVSTTLRYGFLLLMKHPEVEAKVHEEIDRVIGKNRQPKFEDRAKMPYMEAVIHEIQRFGDVIPMSLARRVKKDTKFRDFFLPKGTEVYPMLGSVLRDPSFFSNPQDFNPQHFLNEKGQFKKSDAFVPFSIGKRNCFGEGLARMELFLFFTTVMQNFRLKSSQSPKDIDVSPKHVGFATIPRNYTMSFLPR
|
|||||||
| Gene Name | CYP2A6 | ||||||||
| Uniprot ID | CP2A6_HUMAN | ||||||||
| KEGG Pathway | hsa:1548 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
Exhibits a high coumarin 7-hydroxylase activity. Can act in the hydroxylation of the anti-cancer drugs cyclophosphamide and ifosphamide. Competent in the metabolic activation of aflatoxin B1. Constitutes the major nicotine C-oxidase. Acts as a 1,4-cineole 2-exo-monooxygenase. Possesses low phenacetin O-deethylation activity.
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Mechanism 2 | CYP450 2E1 substrate | CYP450 2E1 inducer | |||||||
| Key Mechanism Factor 2 | |||||||||
| Factor Name | Cytochrome P450 2E1 |
×
Structure
Sequence
MSALGVTVALLVWAAFLLLVSMWRQVHSSWNLPPGPFPLPIIGNLFQLELKNIPKSFTRLAQRFGPVFTLYVGSQRMVVMHGYKAVKEALLDYKDEFSGRGDLPAFHAHRDRGIIFNNGPTWKDIRRFSLTTLRNYGMGKQGNESRIQREAHFLLEALRKTQGQPFDPTFLIGCAPCNVIADILFRKHFDYNDEKFLRLMYLFNENFHLLSTPWLQLYNNFPSFLHYLPGSHRKVIKNVAEVKEYVSERVKEHHQSLDPNCPRDLTDCLLVEMEKEKHSAERLYTMDGITVTVADLFFAGTETTSTTLRYGLLILMKYPEIEEKLHEEIDRVIGPSRIPAIKDRQEMPYMDAVVHEIQRFITLVPSNLPHEATRDTIFRGYLIPKGTVVVPTLDSVLYDNQEFPDPEKFKPEHFLNENGKFKYSDYFKPFSTGKRVCAGEGLARMELFLLLCAILQHFNLKPLVDPKDIDLSPIHIGFGCIPPRYKLCVIPRS
|
|||||||
| Gene Name | CYP2E1 | ||||||||
| Uniprot ID | CP2E1_HUMAN | ||||||||
| KEGG Pathway | hsa:1571 | ||||||||
| Protein Family | Cytochrome P450 family | ||||||||
| Protein Function |
A cytochrome P450 monooxygenase involved in the metabolism of fatty acids (PubMed:10553002, PubMed:18577768). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:10553002, PubMed:18577768). Catalyzes the hydroxylation of carbon-hydrogen bonds. Hydroxylates fatty acids specifically at the omega-1 position displaying the highest catalytic activity for saturated fatty acids (PubMed:10553002, PubMed:18577768). May be involved in the oxidative metabolism of xenobiotics (Probable).
Click to Show/Hide
|
||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of methoxyflurane with CYP450 2E1 and/or 2A6 inducers should generally be avoided. | ||||||||
