Details of Drug-Drug Interaction
| Drug General Information (ID: DDIE1YRW2S) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Cladribine | Drug Info | Flucytosine | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antineoplastics | Antifungal Agents | |||||||
| Structure | |||||||||
| Mechanism of Cladribine-Flucytosine Interaction (Severity Level: Major) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
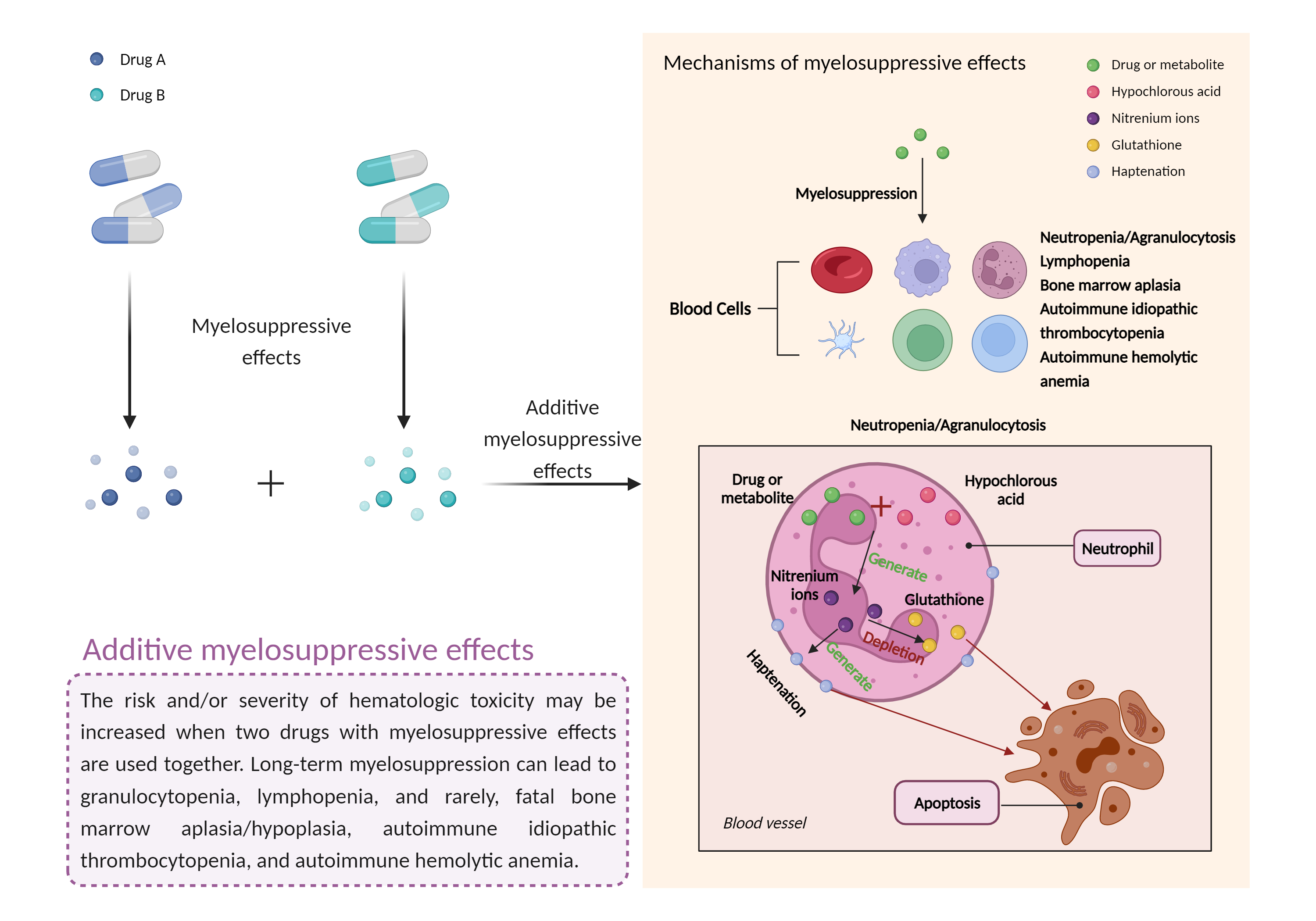
| Additive myelosuppressive effects Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Cladribine | Flucytosine | |||||||
| Mechanism | Myelosuppressive effects | Myelosuppressive effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Myelosuppressive effects | ||||||||
| Factor Description | Myelosuppression, also known as bone marrow suppression, is a decrease in bone marrow activity that leads to a decrease in the production of blood cells. Some blood cell disorders include: erythrocytopenia (anemia), leukopenia (neutropenia), and thrombocytopenia (thrombocytopenia). | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Concomitant use of oral cladribine with immunosuppressive or myelosuppressive agents should be avoided if possible. Acute short-term therapy with corticosteroids can be administered. Caution is advised if IV cladribine must be used in patients who have recently received or are receiving treatment with other immunosuppressive or myelosuppressive drugs, and vice versa. Close clinical and laboratory monitoring for the development of severe hematologic adverse effects is recommended both during and after discontinuation of therapy. In patients who have previously been treated with immunomodulatory or immunosuppressive drugs, consider potential additive effect, mode of action, and duration of effect of the other drugs prior to initiation of cladribine. Patients should be advised to contact their physician if they develop signs and symptoms of infection such as fever, chills, diarrhea, sore throat, muscle aches, shortness of breath, blood in phlegm, weight loss, red or inflamed skin, body sores, and pain or burning during urination. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||||||||||||||||
| 2 | Product Information. Leustatin (cladribine). Ortho Biotech Inc, Raritan, NJ. | ||||||||||||||||||
