Details of Drug-Drug Interaction
| Drug General Information (ID: DDICUTLH1N) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Bacillus calmette-guerin substrain tice live antigen | Drug Info | Human vaccinia virus immune globulin | Drug Info | |||||
| Drug Type | Vaccine | Polyclonal antibody | |||||||
| Therapeutic Class | Vaccine | Immune Globulins | |||||||
| Mechanism of Bacillus calmette-guerin substrain tice live antigen-Human vaccinia virus immune globulin Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
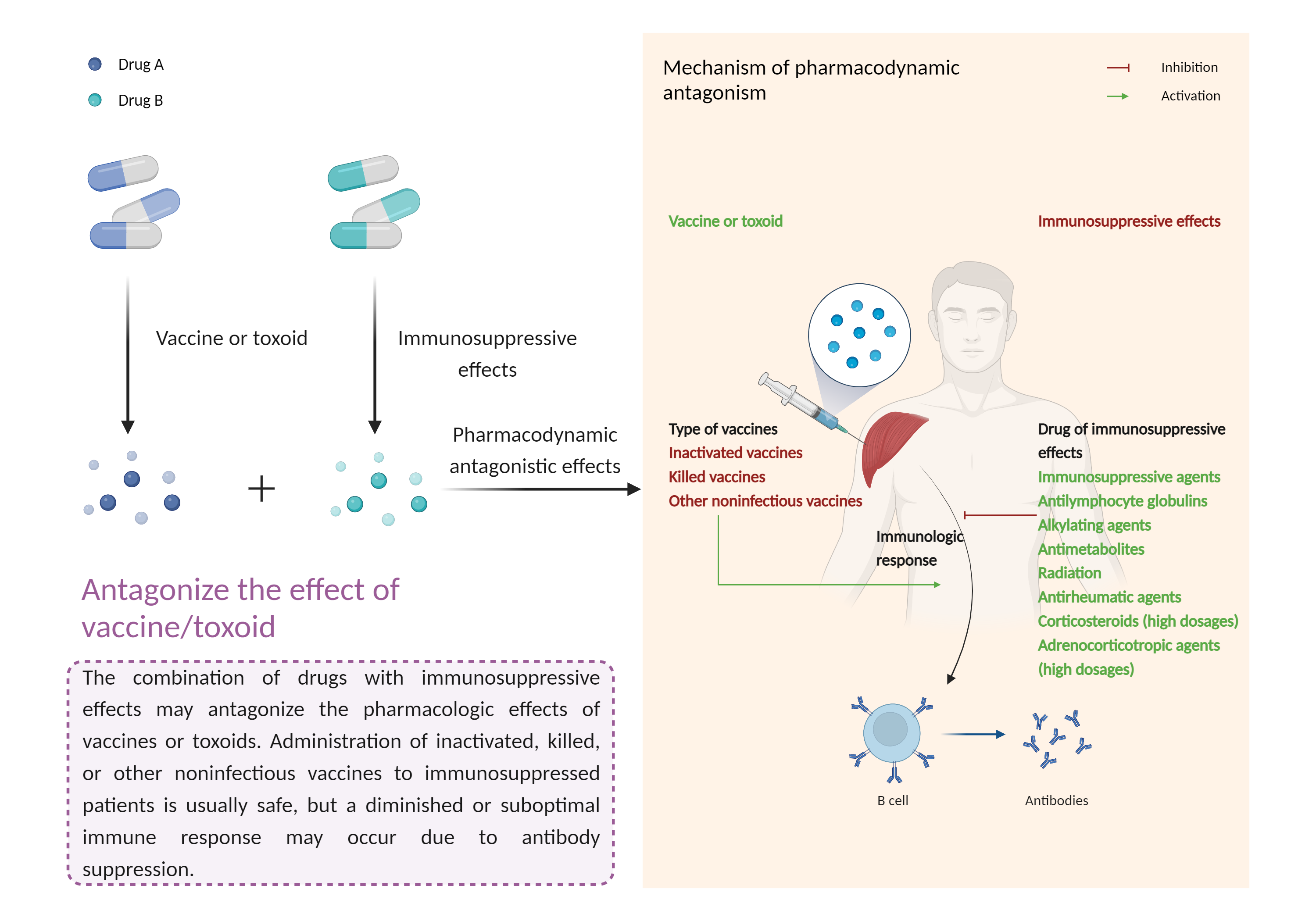
| Antagonize the effect of vaccine/toxoid Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Bacillus calmette-guerin substrain tice live antigen | Human vaccinia virus immune globulin | |||||||
| Mechanism | Vaccine or toxoid | Immunomodulatory effects | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Vaccine/toxoid | ||||||||
| Factor Description | The beneficial effects of the vaccine/toxinoid may be reduced, leading to a diminished or suboptimal immune response. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Immunization with live vaccines should be deferred until approximately 6 months after administration of vaccinia immune globulin intravenous (VIGIV). If such vaccinations are given shortly before or after VIGIV therapy, a revaccination may be necessary. An additional dose of live measles, mumps, or rubella vaccine is usually not needed if the vaccine was administered at least two weeks prior to immune globulin administration. | ||||||||
