Details of Drug-Drug Interaction
| Drug General Information (ID: DDICU0R4QS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Trihexyphenidyl | Drug Info | Levodopa | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Antiparkinson Agents | Dopaminergic Antiparkinsonism Agents | |||||||
| Structure | |||||||||
| Mechanism of Trihexyphenidyl-Levodopa Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
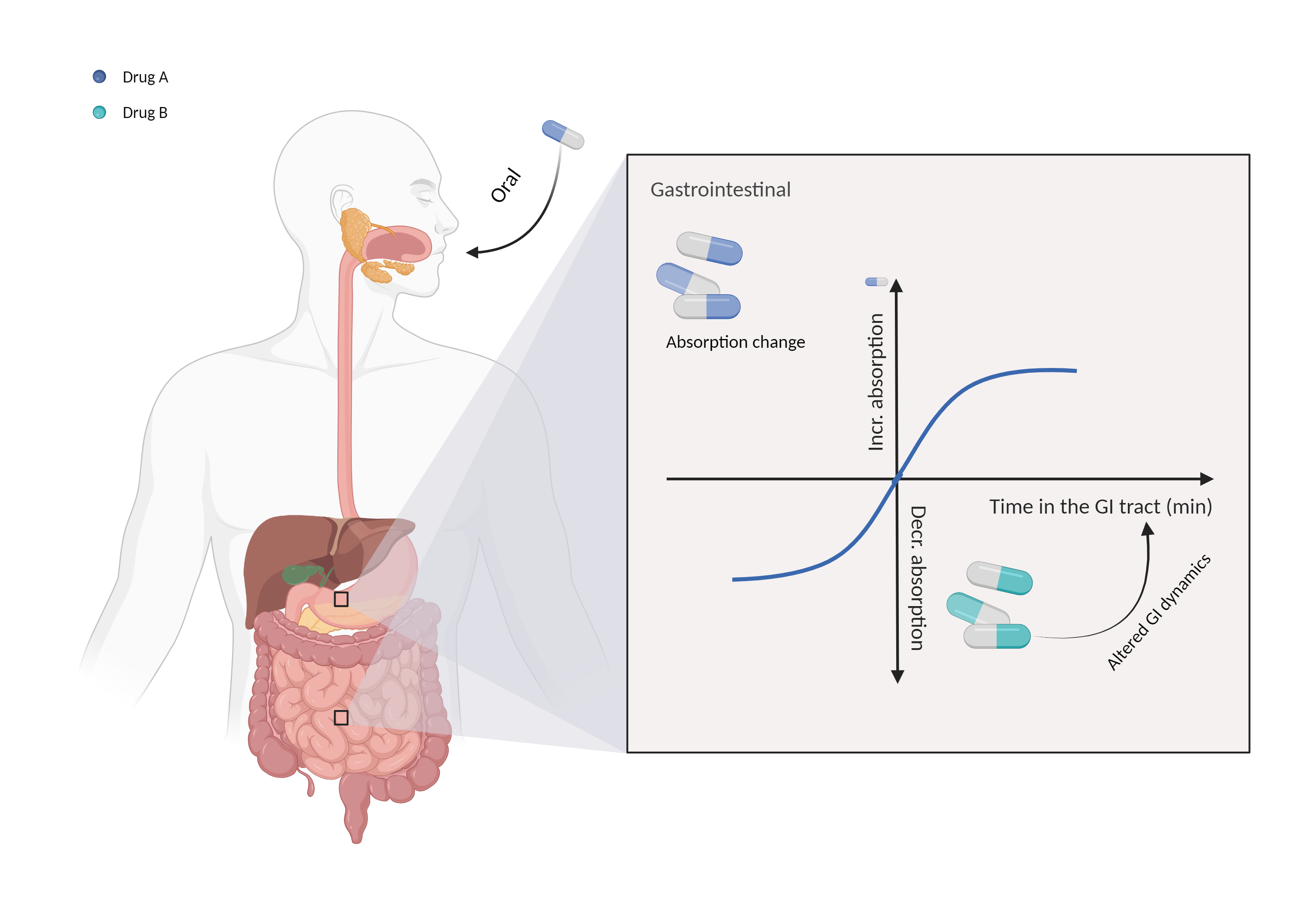
| Altered gastrointestinal dynamics Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Trihexyphenidyl | Levodopa | |||||||
| Mechanism | Delayed gastric emptying | Absorption affected by delayed gastric emptying | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Gastrointestinal Motility | ||||||||
| Factor Description | Gastrointestinal motility is an important factor in determining the absorption of orally administered drugs, and it controls the residence time of the drug in the digestive tract. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | The dosage of both drugs may require reduction. The manufacturer recommends total daily trihexyphenidyl doses of 3 to 6 mg when given concurrently with levodopa. Patients should be monitored for symptom control and side effects. | ||||||||
