Details of Drug-Drug Interaction
| Drug General Information (ID: DDIC9HO8Q0) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Drug Name | Triamterene | Drug Info | Aliskiren | Drug Info | |||||
| Drug Type | Small molecule | Small molecule | |||||||
| Therapeutic Class | Diuretics | Antihypertensive Agents | |||||||
| Structure | |||||||||
| Mechanism of Triamterene-Aliskiren Interaction (Severity Level: Moderate) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
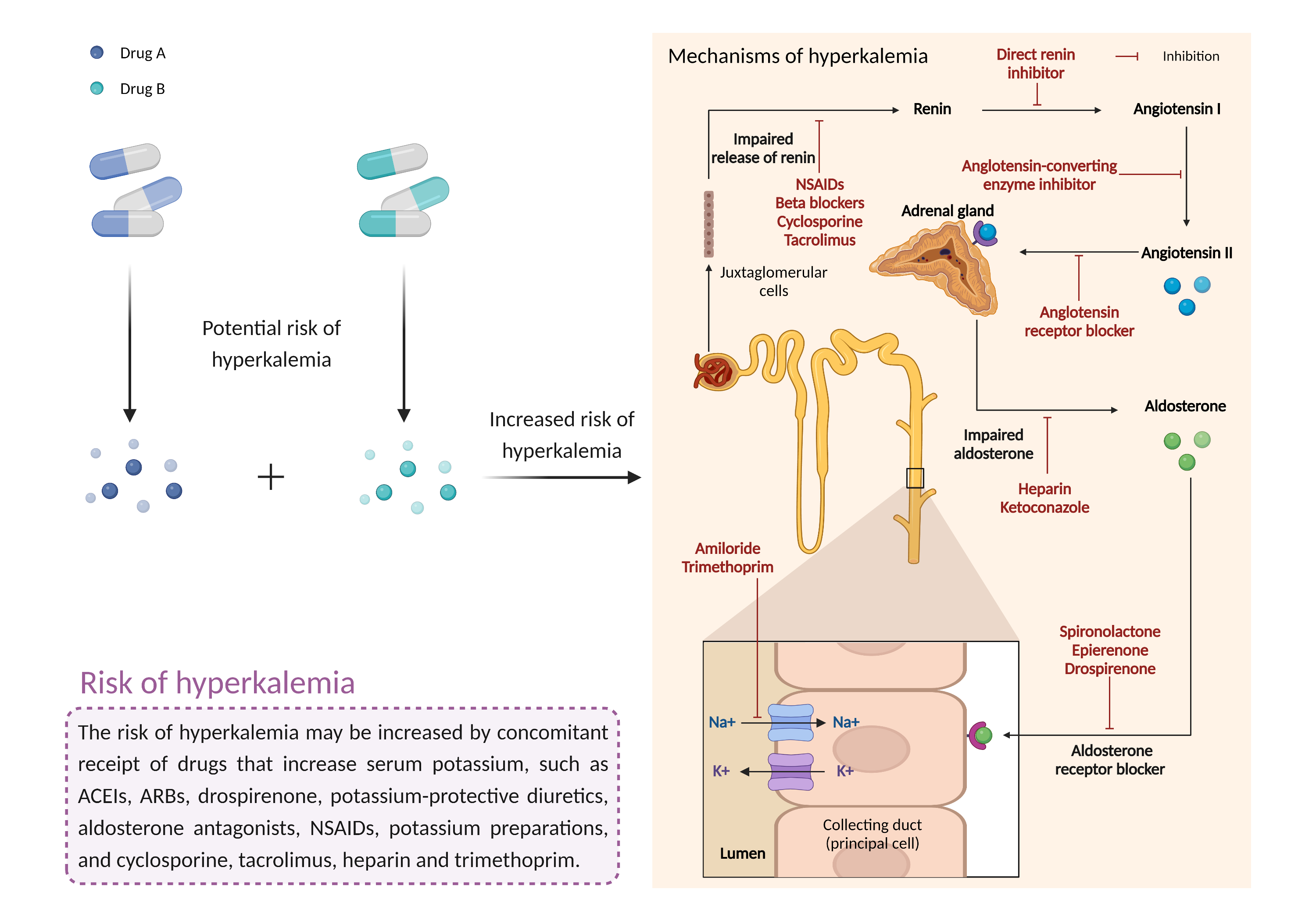
| Increased risk of hyperkalemia Click to Show/Hide Mechanism Graph | |||||||||
 |
|||||||||
| Drug Name | Triamterene | Aliskiren | |||||||
| Mechanism | Hyperkalemia | Hyperkalemia | |||||||
| Key Mechanism Factor 1 | |||||||||
| Factor Name | Hyperkalemia | ||||||||
| Factor Description | Hyperkalemia is a condition in which the level of potassium in the blood is higher than normal. While mild cases may not produce symptoms, severe hyperkalemia can lead to fatal arrhythmias if left untreated. | ||||||||
| Mechanism Description |
|
||||||||
| Recommended Action | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Management | Caution is advised if aliskiren is used in combination with potassium-sparing diuretics or potassium salts. Routine monitoring of electrolytes and renal function may be indicated, particularly in patients with renal impairment, diabetes, old age, worsening heart failure, or a risk for dehydration. Patients should be advised to seek medical attention if they experience signs and symptoms of hyperkalemia such as weakness, listlessness, confusion, tingling of the extremities, and irregular heartbeat. | ||||||||
| References | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Product Information. Tekturna (aliskiren). Novartis Pharmaceuticals, East Hanover, NJ. | ||||||||||||||||||
